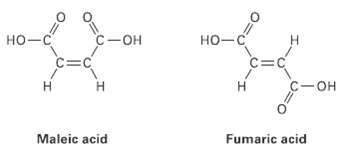
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the
Question:
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain
Transcribed Image Text:
с — он но-с C-OH но-с C=C c=C С —он Maleic acid Fumaric acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
In maleic acid the individual dipole moments ad...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A water molecule has a dipole moment of 6.3 x 10 30 C m. A sample contains 1021 molecules of water. Their dipole moments are all oriented in the direction of an electric field of 2.5 x 10 N/C....
-
The PF3 molecule has a dipole moment of 1.03 D, but BF3 has a dipole moment of zero. How can you explain the difference?
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
A bank reconciliation takes time and must balance. An employee was struggling in balancing the bank reconciliation. Her supervisor told her to plug (make an unsupported entry for) the difference,...
-
Elaborate rules exist that require employers to prepay various types of Federal taxes. Summarize the major issues that an employer must resolve if it is to comply with the requirements?
-
Table 2.9 gives data on mean Scholastic Aptitude Test (SAT) scores for collegebound seniors for 19722007. These data represent the critical reading and mathematics test scores for both male and...
-
For each of the following situations, calculate the \(t\)-statistic \((t)\) : a. \(\mathrm{X}^{-}=11.00 ; \mu=5 ; s \mathrm{X}^{-}=3.00\) b. \(X^{-}=26.00 ; \mu=31 ; s X^{-}=2.00\) c. \(X^{-}=19.60 ;...
-
Suppose Ramirez Ltd. Just issued a dividend of $1.89 per share on its common stock. The company paid dividends of $1.47, $1.62, $1.67, and $1.78 per share in the last four years. If the stock...
-
1. For Firm G, compute cost of direct labor used. 2. For Firm G, compute cost of direct materials used. 3. For Firm G, compute cost of goods manufactured. 4. For Firm G, compute cost of goods sold....
-
You are the auditor of Vegatron Services Inc., a privately owned full-service cleaning company following ASPE that is undergoing its first audit for the period ending September 30, 2011. The bank has...
-
Assign formal charger to the atoms in each of the followingmolecules: ( NNEN: CH (c) HCN3DN-DN: (a) H3C-N-O: CH
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
Delineate the value-maximizing motives for mergers. How are these motives interrelated?
-
List three (3) examples of the tools and equipment that are used for partial highlighting and foiling.
-
What is the term for the separation of a service from the entity (or entities) providing that service? Explain.
-
How can the course of Criminal Law enhance your professional success? How can you apply what you learned in this criminal law to a future career as a autopsy technition?
-
What are some excellent memory techniques for Criminal Procedure (law school), not criminal law?
-
The Maybe Pay Life Insurance Co. is trying to sell you an investment policy that will pay you and your heirs $32,000 per year forever. Suppose a sales associate told you the policy costs $477,000. At...
-
Give some examples of actions that would probably fall within the conduct proscribed by (a) extortion and (b) vandalism or malicious mischief statutes.
-
Continuation of Exercise 4-83. (a) What is the probability that the first major crack occurs between 12 and 15 miles of the start of inspection? (b) What is the probability that there are no major...
-
a. Show that b. Use the result of part (a) to show that for a stable system at equilibrium ( V /T) S and ( V /T) P must have opposite signs. c. Two separate measurements are to be performed on a gas...
-
Draw all of the stereo isomers of 1, 2-dimethylcyclopropane. Explain which rotate plane-polarized light.
-
Explain whether these compounds rotate plane-polarized light: H a) HC-CCH, HC Br c) Br J. s CI b) Cl CI d) HC -CH3
-
Draw Fischer projections for these compounds? CHOH a) H-C-CI CH3 COH b) HC-OH A CH3 c) H CHCH CH CH3
-
Supply is the connection between the cost of a decent and the amount ready to move costing that much. It very well might be addressed as a table or diagram relating cost and amount provided. Makers,...
-
If 50% of the men 18-34 years old watch television on Mondays at 11 PM, and 30% of those viewers watch Monday Night Football, what is Monday Night Football's rating among men 18-34?
-
Assume you are a media planner and you have just completed a media plan using both magazine and television advertising. Your magazine schedule reaches 50 percent of the target audience and your...

Study smarter with the SolutionInn App


