Method of continuous variation. Make a graph of absorbance versus mole fraction of thiocyanate from the data
Question:
Method of continuous variation. Make a graph of absorbance versus mole fraction of thiocyanate from the data in the table.
.png)
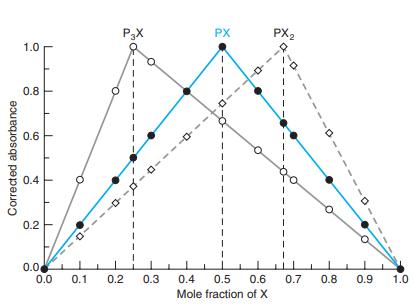
(a) What is the stoichiometry of the predominant Fe(SCN)n3-n species?
(b) Why is the peak not as sharp as those in Figure 18-8?
(c) Why does one solution contain 10.0 mM acid and the other 15.0 mM acid?
Figure 18-8
Transcribed Image Text:
mL Fe3 solution 30.00 27.00 24.00 21.00 18.00 15.00 12.00 9.00 6.00 3.00 mL SCN solution 3.00 6.00 9.00 12.00 15.00 18.00 21.00 24.00 27.00 30.00 Absorbance at 455 nm 0.001 0.122 0.226 0.293 0.331 0.346 0.327 0.286 0.214 0.109 0.002 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a Maximum absorbance occurs at XSCN 0500 stoichiometry 1 1 ...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Simulating a Job's plot. Consider the reaction , for which K [AB 2 ]/[A][B ]2 . Suppose that the following mixtures of A and B at a fixed total concentration of 104M are prepared: (a) Prepare a...
-
Gold nanoparticles (Figure 16-29) can be titrated with the oxidizing agent TCNQ in the presence of excess of Br to oxidize Au(0) to AuBr in deaerated toluene. Gold atoms in the interior of the...
-
The variation of refractive index, n, with wavelength for fused silica is given by where is expressed in μm. (a) Make a graph of n versus λ with points at the following...
-
Suppose a town concludes that it costs on average $30.00 per household to manage the disposal of the waste generated by households each year. It is debating two strategies for funding this cost: (1)...
-
Jill Epp, a friend of yours, overheard a discussion at work about changes her employer wants to make in accounting for uncollectible accounts. Jill knows little about accounting, and she asks you to...
-
In Problem S8-2, a time study was conducted for the job of sewing baseball-style caps. Using a sample standard deviation of 0.25, determine the number of cycles for the time study such that the...
-
What are some of the advantages and disadvantages of using online recruitment?
-
Do you think global businesses would be willing to subscribe to a global code of conduct? Explain your answer.
-
how do you write $ 3 , 4 5 1 in financial notation
-
Adden Company signs a lease agreement dated January 1, 2019, that provides for it to lease non-specialized heavy equipment from Scott Rental Company beginning January 1, 2019. The lease terms,...
-
Now we use Solver to find K for the previous problem. The only absorbing species at 332 nm is the complex, so, from Beer's law, [complex] = A/ (because pathlength = 1.000 cm). I 2 is either free or...
-
The indicator xylenol orange (Table 11-3) forms a complex with Zr(IV) in HCl solution. Prepare a Job plot from the data in the table and suggest the stoichiometry of the complex (xylenol orange) x Zr...
-
A famous study by Amos Tversky and Nobel laureate Daniel Kahneman asked people to consider two hospitals. Hospital A is small and has 15 babies born per day. Hospital B has 45 babies born each day....
-
Exactly 205 girls. Assume that 400 births are randomly selected. Use subjective judgment to determine whether the given outcome is unlikely, and also determine whether it is unusual in the sense that...
-
Exactly 111 girls. Assume that 400 births are randomly selected. Use subjective judgment to determine whether the given outcome is unlikely, and also determine whether it is unusual in the sense that...
-
It is impossible to pass a statistics test without studying. Express the indicated degree of likelihood as a probability value between 0 and 1.
-
Describe the three types of share-based payment transactions under IFRS 2.
-
One form of the rule of complements is this: P(A or A) = 1. Write a sentence describing the message that this rule represents.
-
Which astronomical objects that appear as points of light to the unaided eye appear as disks with the help of a telescope?
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Calculate the concentration of H 2 Y 2- at the equivalence point in Exercise 11-C. Exercise 11-C Calculate pCu 2+ (to the 0.01 decimal place) at each of the following points in the titration of 50.0...
-
Suppose that 0.010 0 M Mn 2+ is titrated with 0.005 00 M EDTA at pH 7.00. (a) What is the concentration of free Mn 2+ at the equivalence point? (b) What is the quotient [H 3 Y - ]/[H 2 Y 2- ] in the...
-
Suppose that 0.010 0 M Mn 2+ is titrated with 0.005 00 M EDTA at pH 7.00. (a) What is the concentration of free Mn 2+ at the equivalence point? (b) What is the quotient [H 3 Y - ]/[H 2 Y 2- ] in the...
-
Blue Company changed depreciation methods in 2025 from double-declining balance to straight-line. Depreciation prior to 2025 under double-declining-balance was 592,400, whereas straight-line...
-
Figure out, salaries, net income, add net income with retained earnings and totally stockholder equity , ?accounts payable and total liabilities and stockholders equity RILEY, INCORPORATED Statement...
-
Determine the material inventory balance at the end of may? Received Issued Receiving Received Materials Report Number Received Quantity Unit Price Requisition Number Issued Quantity Issued Balance...

Study smarter with the SolutionInn App


