One million pound-moles per day of a gas of the following composition is to be absorbed by
Question:
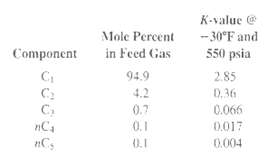
One million pound-moles per day of a gas of the following composition is to be absorbed by n-heptane at -30?F and 550 psia in an absorber having 10 theoretical stages so as to absorb 50% of the ethane. Calculate the required flow rate of absorbent and the distribution, in lbmol/h, of all the components between the exiting gas and liquid streams.
Transcribed Image Text:
K-value @ Mole Percent -30°F and 550 psia Component in Feed Gas 94.9 2.85 D. 36 0.066 C2 C, nC4 4.2 0.7 0.1 0.017 0.1 0.004
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
From Eq 548 Solving and using Eq 538 Therefore L 0 0500361000000 180000 lbmolday To compute the dis...View the full answer

Answered By

Pallab Nath
I am pallab nath.I have completed my Masters in chemistry from University of Kalyani with an overall percentage of 75.31%. I have also participated in a project which is focused on "synthesis of ruthenium metal complexes".
I have a very decent knowledge in every fundamental branch of chemistry(inorganic,physical,organic).I love to solve various problems from chemistry.I can teach many fundamental concepts from chemistry in a very interesting way and help the students to understand and solve the problems from different concept in chemistry.Besides, I can help the students by providing them with perfect conception,Problem solving methods and applications in various branches from chemistry.Overall i can say that I am quite capable of helping the students to understand and solve every questions from chemistry and have a better concept in chemistry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The pressure of a gas at the triple point of water is 1.35 atm. If its volume remains unchanged, what will its pressure be at the temperature at which CO2 solidifies?
-
At what temperature of a gas will the number of molecules, whose velocities fall within the given interval from v to v + dv, be the greatest? The mass of each molecule is equal to m.
-
A gas obeys the following equation of state: P(v-b) = RT where the constant b was found to be 0.2 m 3 /kmol. Over the temperature range 300-400 K the heat capacity may be taken to be constant at 40...
-
We often speak of how price rations goods. What are other rationing measures in clinics in which free care is provided?
-
The following information is available for PepsiCo, Inc. (in U.S. millions): In the notes to its financial statements, PepsiCo disclosed that it uses the FIFO and average cost formulas to determine...
-
In Exercise, determine the measure of x. 57 27
-
In Example 17.11, assume that the promised amounts to \(A\) and \(B\) are \(\$ 110\) and \(\$ 90\), respectively. Develop the new table of results. What are the prices of \(A\) and \(\mathrm{B}\) ?...
-
St. Lukes Medical Center has a single operating room that is used by local physicians to perform surgical procedures. The cost of using the operating room is accumulated by each patient procedure and...
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?
-
Linda Larue has arthritis. Her chiropractor advised her that she needed to swim daily to alleviate her pain and other symptoms. Consequently, Linda and her husband, Philo, purchased for $ 400,000 a...
-
Solve Example 5.3 with the addition of a heat exchanger at each stage so as to maintain isothermal operation of the absorber at (a) 125F (b) 150oF What is the effect of temperature on absorption in...
-
A stripper operating at 50 psia with three equilibrium stages is used to strip 1,000 kmol/h of liquid at 300F having the following molar composition: 0.03% C1, 0.22% C2, 1.82% C3, 4.47% nC4, 8.59%...
-
Consider the two-dimensional Poiseuille flow of Prob. 499. The fluid between the plates is water at 40C. Let the gap height h = 1.6 mm and the pressure gradient dP/dx = 230 N/m 3 . Calculate and plot...
-
A program is to read a numeric score (0 to 100) and display an appropriate letter grade (A, B, C, D, or F). 1. What is the functional domain of this program? 2. Is exhaustive data coverage possible...
-
The C++ thread library provides a function that returns the number of threads that the hardware is capable of running. Modify the parallel merge sort so that the user specifies a minimum chunk size....
-
Write a queue application that determines if two files are the same.
-
Why is it good practice to put a class declaration in one file and the implementation in another?
-
Differentiate between top-down and bottom-up integration testing.
-
Universal Oil and Gas Company has been using full cost accounting for 20 years. The following data relate to US properties as of 12/31/20. REQUIRED: Compute DD&A using the unit-of-revenue method....
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
Outline the key differences between how people learn.
-
Within a porous particle, why are mass and heat transfer not analogous?
-
Ion exclusion is a process that uses ion-exchange resins to separate nonionic organic compounds from ionic species contained in a polar solvent, usually water. The resin is presaturated with the same...
-
What are the four steps that occur during the adsorption of a solute from a gas or liquid mixture? How do they affect adsorptive bandwidth, resolution, and throughput? Which step may be almost...
-
What is one way in which a leader can apply the "Status" part of the SCARF model to trigger the "Reward" response?
-
Complete the table to assess risks and develop contingencies for resources you are planning for. Potential risks Planned Response Risk associated with the acquisition or implementation of the...
-
Assignment: persuasive essay for or against Andrew Jackson. In you essay answer the question "Was he a "people's president" or was he more like a king?" Documentary:...

Study smarter with the SolutionInn App


