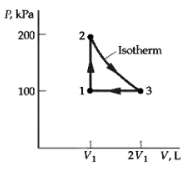
One mole of an ideal monatomic gas at an initial volume V1 = 25 L follows the
Question:
One mole of an ideal monatomic gas at an initial volume V1 = 25 L follows the cycle shown in figure. All the processes are quasi-static. Find
(a) The temperature of each state of the cycle,
(b) The heat flow for each part of the cycle, and
(c) The efficiency of thecycle.
Transcribed Image Text:
P, kP | 200 2. Isotherm 100 2V1 V,L V1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a Using PV nRT T 1 100 258314 K 3007 K T 2 T 3 6014 K b Q 1...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
Consider one mole of an ideal monatomic gas at 300K and 1 atm. First, let the gas expand isothermally and reversibly to twice the initial volume, second, let this be followed by an isentropic...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
A heat engine follows the cycle shown in the figure. (a) How much net work is done by the engine in one cycle? (b) What is the heat flow into the engine per cycle? 4.00 atm 1.00 atm 0.200 m 0.800 m3 T
-
In Exercises find the indefinite integral. sech(2x1) dx
-
Data concerning a recent period's activity in the Assembly Department, the first processing department in a company that uses the FIFO method in its process costing, appear below: A total of 26,000...
-
Griffin and Lasky, Inc. (G&L), supplies industrial automation equipment and machine tools to the automotive industry. G&L recognizes revenue on its long-term contracts over time. Customer orders have...
-
What is the difference between domain definition and domain dictionary? Give an example for each.
-
Prepare a performance report for Imperial Data Devices using the budget information from Exercise 10-15 and the next performance information. In Exercise 10-15, Expected manufacturing costs for...
-
How do ethical leaders demonstrate authenticity and ethical authenticity, aligning personal values with organizational goals, and modeling ethical behavior to cultivate ethical awareness and...
-
Selected financial information (in 000s) from the financial statements of Moosomin Ltd. for Years 6 and 7 is as follows: Required (a) Convert Moosomins financial statements for both Year 6 and Year...
-
An engine using 1 mol of an ideal gas initially at V 1 = 24.6 L and T = 400 K performs a cycle consisting of four steps: (1) an isothermal expansion at T = 400 K to twice its initial volume, (2)...
-
An ideal gas (? = 1.4) follows the cycle shown in figure. The temperature of state 1 is 200 K. Find (a) The temperatures of the other three states of the cycle and (b) The efficiency of the cycle. P,...
-
During the week of February 9-15, 2001, the U.S. rose market cleared at a price of $1.00 per stem, and 4,000,000 stems were sold that week. During the week of June 5-11, 2001, the U.S. rose market...
-
How does a universal bank common in some other parts of the world compare with a commercial bank in the United States? How does the U.S. holding company structure compare with a universal bank?
-
What is meant by systemic risk? How is this related to economic externalities? How is systemic risk being addressed by the regulatory authorities?
-
China is experiencing inflationary pressures. How is this related to its exchange rate policy? Is its currencythe yuanundervalued or overvalued? Why has China raised its reserve requirements on...
-
What must a country with a fixed exchange rate do to hit its target for the exchange rate if its currency is undervalued? Overvalued? Is the country also able to pursue other goals with monetary...
-
Could a large decline in asset prices occur without prompting a financial crisis? Any examples?
-
Why is hedging transaction exposure generally warranted?
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
Under what conditions will a bond sell at a premium above par? At a discount from par?
-
The electric field at a distance of 0.145 m from the surface of a solid insulating sphere with radius 0.355 m is 1750 N/C. (a) Assuming the sphere's charge is uniformly distributed, what is the...
-
A conductor with an inner cavity, like that shown in Fig. 22.23c, carries a total charge of + 5.00 nC. The charge within the cavity, insulated from the conductor, is -6.00 nC. How much charge is on...
-
Apply Gauss's law to the Gaussian surfaces S2, S3, and S4 in Fig. 22.21b to calculate the electric field between and outside the plates.
-
The quality-control department of Starr Communications, the manufacturer of video-game DVDs, has determined from records that 1.6% of the DVDS sold have video defects, 0.9% have audio defects, and...
-
What is the half-life in hours of procainamide in a patient whose total clearance is estimated to be 20 L/h and the volume of distribution is 120 L assuming one compartment, open model, first- order...
-
1. The technologies, such as ChatGPT, has been launched before introducing even a baseline of safety measures. This goes back to trade-offs that companies make, and people just watch the impact from...

Study smarter with the SolutionInn App


