Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason
Question:
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount.
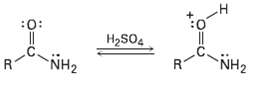
Transcribed Image Text:
:0: H2SO4 NH2 NH2 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
0 NH3 NH NProtonation ...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An equimolar mixture of oxygen and nitrogen enters a compressor operating at steady state at 10 bar, 220 K with a mass flow rate (m) of 1 kg/s. The mixture exits the compressor at 60 bar, 400 K with...
-
A friend of yours has performed three titrations: strong acid with a strong base, weak acid with a strong base, and weak base with a strong acid. He hands you the three titration curves, saying he...
-
Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown here) which shows that the two carbon-carbon bonds an identical bond order of 1.5 and that the unshared...
-
You recently joined an accounting consulting firm. Your first client is a new manufacturing start-up that is trying to set up their costing system, and develop good processes for their upcoming...
-
What would you describe as Costco's basic strategy as a retailer? How do human resource practices support that strategy?
-
What is the distinction between glass transition temperature and melting temperature?
-
State the advantages of a CRSA system for: (a) company managers (b) risk managers (c) internal auditors (d) companies as a whole.
-
Farrior Fashions needs to replace a beltloop attacher that currently costs the company $40,000 in annual cash operating costs. This machine is of no use to another company, but it could be sold as...
-
Tarrant Amertex Regional Hospital, one of our hospitals in the Fort Worth area, has been in need of a new Chief Executive Officer. Last week, we reached a "handshake" dealthough per current protocols...
-
In 2018, Kida Company purchased more than $10 million worth of office equipment 11-6, 11-9 under its special ordering system, with individual orders ranging from $5,000 to $30,000. Special orders...
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
Air at 20oC, 95 kPa is compressed in a piston-cylinder device of volume 1 L in a frictionless and adiabatic manner. If the volumetric compression ratio is 10, determine (a) The final temperature. (b)...
-
Solve the Following: Given f: WIR Define n2"-1 fn (w) = [ 2 (w/w) ) +n1(w//(w)n) k=0 1. Show fn is a simple function 2. Show that these sets form a partition of
-
What are the three main principles applied to corporate lending proposals? Q2. The five Cs is one method of structuring a loan approval process. What fundamental piece of information does it ignore...
-
Use the below capitalization table for Startup Inc. (Startup) and assume that: Founders (Natalie & Matt) purchased their shares of common stock for $0.001 per share (for a total of $3,500). A...
-
Form 706 706 United States Estate (and Generation-Skipping Transfer) Tax Return (Rev. August 2021) Estate of a citizen or resident of the United States (see instructions). To be filed for OMB No....
-
The 16 th tab in your Financial Statement Analysis Template must be labeled Projected Income Statements. Three years of projected statements should be included, starting with the most recent year...
-
You are offered \($200\) now plus \($100\) a year from now for your used computer. Since the sum of those two amounts is \($300\), the buyer suggests simply waiting and giving you \($300\) a year...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Write the formula for the conjugate acid of each base. a. NH3 b. CIO C. HSO4 2- d. CO3-
-
As noted in the "A Word about . . . Methane, Marsh Gas, and Miller's Experiment" on page 60, methane can be formed in muddy sediments because of the reducing environment (i.e., lack of oxygen). Write...
-
Explain why 1,3-difluorobutane is a correct IUPAC name, but 1,3-dimethylpentane is not a correct IUPAC name.
-
Draw Newman projections for two different staggered conformations of butane (looking end-on at the bond between carbon-2 and carbon-3), and predict which of the two conformations is more stable. (If...
-
Question 1. From the research for this terms Crisis Management Team Assignment - Taylor swift and Ticket Master Crisis a. Explaining, with some detail, three (3) specific things that the leader(s) in...
-
If individual values that underline ethics are developed at a young age, what might this suggest about the potential for ethical conflicts to arise within an organization?
-
What might be challenges for public administrators in orchestrating ethical performances when working with individual ethics inside and outside the organization?

Study smarter with the SolutionInn App


