Question: Predict the approximate chemical shifts, multiplicities and integrals for the absorptions in the 1H-NMR spectra of thesecompounds: CH2 b) .- COCH CH3 a) d) CH;CHCH
Predict the approximate chemical shifts, multiplicities and integrals for the absorptions in the 1H-NMR spectra of thesecompounds:
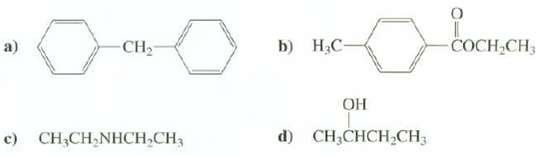
CH2 b) .- COCH CH3 a) d) CH;CHCH CH3 c) CH;CH,NHCH,CH3
Step by Step Solution
3.51 Rating (168 Votes )
There are 3 Steps involved in it
a chemical shift multiplicity integral c 78 23 ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (81).docx
120 KBs Word File


