Predict the multiplicities of the absorptions for the hydrogen's of these groups, assume that hydrogen's labeled a
Question:
Predict the multiplicities of the absorptions for the hydrogen's of these groups, assume that hydrogen's labeled a are different from those labeled x but that all of those labeled a are identical and all of those labeled x are identical.
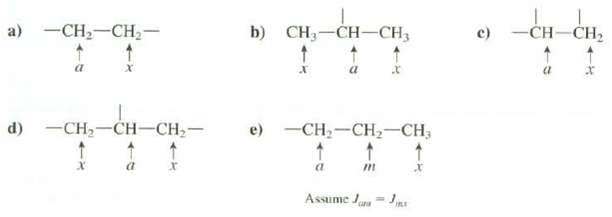
Transcribed Image Text:
a) -CH,-CH2- -CH-CH2 b) CH3-CH-CH3 c) e) -CH,-CH2-CH; -CH,-CH-CH,- d) т Assume Juna = Jn LE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
In general the absorption peak for ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the multiplicities of the indicated hydrogen's in the 1H-NMR spectra of these compounds: CH,CI I H I - + CH,CI CI - c) b) --- I H I - ()
-
Predict the characteristic infrared absorptions of the functional groups in the following molecules. (a) Cyclohexene (b) Pentan-2-ol (c) Pentan-2-one (d) Pent-1-yne (e) Diethylamine (f) Pentanoic...
-
All facts about this NFPO are identical to those described in Problem 10, except that the deferral method of recording contributions is used for accounting and for external financial reporting. Fund...
-
Determine the vector A-C, given the vectors A and C in the figure. (Figure 1) Figure B (B=26.5) 56.0% (A = 44.0) 28.0 C(C= 31.0) 1 of 1 Determine the magnitude of the vector A - . Express your...
-
How should Acorn organize now, considering both their commercial business and their growing government business?
-
1. What mistakes do you think John Mitchell made with the way he solved the problem of limited office space? Explain. 2. What approach would you have used if you were Mitchell? Why? 3. What are...
-
How long does it take an ambulance to respond to a request for emergency medical aid? One of the goals of one study was to estimate the response time of ambulances using warning lights (Ho \&...
-
1. What are you (in the role of Kathy Dalton) going to present at the outsourcing strategy meeting, and why? 2. When companies like Dell are contracting outsourcing in some areas, should eBay...
-
Explain what is meant by corporate governance. In September 2014, Alibaba listed on the New York Stock Exchange in what was, at the time, the largest IPO in history. Explain some of the reasons that...
-
Presented below is an aging schedule for Bryan Company at December 31, 2021. At December 31, 2021, the unadjusted balance in Allowance for Doubtful Accounts is a credit of $8,000. Instructions a....
-
Construct a tree diagram for the absorption of Hm assume that Jam . . - -C-
-
Predict the 1H.NMR spectra of these compounds include the approximate chemical shift, multiplicity, and integral for each type ofhydrogen. CI b) CH;CHCH; ) C,CH,H c) CH,CH,OCH,CH3 CH2CH2NO2 f)...
-
Consider each of the following scenarios: a. A seller orally agrees with one of its best customers to deliver goods in exchange for $10,000. While the sellers practice is to obtain a written sales...
-
An 8.24 x 10 -7 C charge feels a 7.89 x 10 -8 N force when it moves 2090 m/s at a 29.4 angle to a magnetic field. How strong is the field?
-
FOOD AND BEVERAGE Description of bread and butter plate: Whats the use of Bread and butter plate? Importance of bread and butter plate:
-
Most Beagles have shoulder heights between 36 and 41 centimeters. The following compound inequality relates the estimated shoulder height (in centimeters) of a dog to the internal dimension of the...
-
The R in the top equitation is Slide #8 is the percentage of the face value expected to be recovered in case of a bankruptcy, e.g., 35%. Absolute valuation (DCF) of corporate bonds After some...
-
Question 1 Based on the cash flow shown over 10 years for three projects, which should be selected using the concept of IRR and Delta IRR? MARR 14% Year Project P Project Q Project R 0 (29.200.00)...
-
Go to http://www.gwdc.org/docs/msesp/Technical%20 Proposal.pdf to see a process for describing and creating a Statement of Need for the Minnesota State Energy Sector Partnership. In your opinion,...
-
A summary of changes in Pen Corporation's Investment in Sam account from January 1, 2011, to December 31, 2013, follows (in thousands): ADDITIONAL INFORMATION 1. Pen acquired its 80 percent interest...
-
In the 1980s, many firms in China were government-sponsored enterprises that were either owned by the government or received large government subsidies. Since the 1990s, the Chinese government has...
-
Classify the following sigma tropic reaction by order [x, y], and tell whether it will proceed with suprafacial or antarafacial stereochemistry: -H H-
-
Propose a mechanism to account for the fact that heating 1-deuterioindene scrambles the isotope label to all three positions on the five-memberedring. 1-Deuterioindene
-
When a 2, 6-disubstituted allyl phenyl ether is heated in an attempted Claisen rearrangement, migration occurs to give the p-allyl product as the result of two sequential pericyclic...
-
How do codes of ethics affect professional, community service delivery? Question 2 Describe the steps you might follow in self-management. Question 3 Conduct appropriate research into the five-factor...
-
You are applying for a job with a community services organization as a communications/ human resources manager. They have asked you to submit a detailed report of what you would do to develop,...
-
Water is being pumped from an open water reservoir at the rate of 2.0 kg/s at 10C to an open storage tank 1500 m away. The pipe used is schedule 40 3 in. pipe and the frictional losses in the system...

Study smarter with the SolutionInn App


