Question: Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain. , , . -C-C-C-
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain.
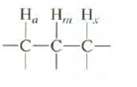
, , . -C-C-C-
Step by Step Solution
★★★★★
3.32 Rating (173 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The signal from H m will appear ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
15-C-O-S (73).docx
120 KBs Word File


