Predict the product of the following Diels-Alderreaction: - C=C
Question:
Predict the product of the following Diels-Alderreaction:
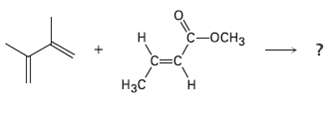
Transcribed Image Text:
С-оснз н C=C Нзс Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Strategy Draw the reactants in an orientation that shows where the new bonds will f...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Predict the product of the following reaction. O (1) BrMg (2) H2O MgBr (1 equiv.)
-
Predict the product of the following reaction:
-
Classify these equations according to their order, degree, type and linearity. Order Degree IV DV Linearity
-
What was the role of the factor in the mid-19th century economy? Does such a role exist in the modern economy?
-
In a balanced three-phase Y-Y system, the source is an abc sequence of voltages and Van = 100 20 V rms. The line impedance per phase is 0.6 + j1.2 , while the per-phase impedance of the load is 10 +...
-
For an equimolar solution of \(n\)-pentane and \(n\)-hexane, compute: (a) The dew-point pressure at \(120^{\circ} \mathrm{F}\) (b) The bubble-point temperature at \(1 \mathrm{~atm}\) (c) The vapor...
-
You are the vice-president of finance of Sandy Alomar Corporation, a retail company. The company prepared two different schedules of gross margin for the first quarter ended March 31, 2008. These...
-
Evaluate the role of non-traditional thermodynamic cycles, such as supercritical CO2 cycles, organic Rankine cycles, and Kalina cycles, in improving energy efficiency and reducing emissions in power...
-
Use the information below and prepare the Multi-Step Income Statement for Hot Place on August 31. Sales $1,250,000 Interest Revenue 25 Sales Discounts 15,000 Sales Returns and Allowances 30,000 Cost...
-
Why do you suppose 1, 4 adducts of 1, 3-butadiene are generally more stable than 1, 2 adducts?
-
Which of the following alkenes would you expect to be good Diels-Alder dienophile?s? (b) (a) H2C=CHCI %3H2H2C (d) (c) (e)
-
Why do you think wages tend to be rigid in the downward direction?
-
5. (a) Write in words how to read the following out loud {n EN n is a prime number less than 10}. (b) Use the set-roster notation to indicate the elements in the set.
-
Describing the closure of a factory in a small town as "reallocation of resources" is an example of 5 pts
-
Make a list of all of the support services (for example: housekeeping, security, dietary services) required in your work setting or setting of interest. Estimate the annual costs of each of these...
-
Create Java Program that solves this question 1. Create 2 interfaces: BankInterface and PetInterface. BankInterface contains only 1 abstract method: accountType() which does not return anything and...
-
Imelda needs to find a way to pay for college next semester. She doesn't want to work more than 20 hours a week because she is taking 15 credit hours. Also, she wants to get some "real world"...
-
a. Find the partial-fraction expansion. b. AVerify (a) by using the residue command in MATLAB. \(\frac{s}{(s+1)\left(s^{2}+2 s+2 ight)}\)
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
What is osmosis? What is osmotic pressure?
-
How would you convert the following compounds to aromatic compounds? (a) (b) (c) (d) (e) (f) Cl
-
An unknown compound gives the following mass, IR, and NMR spectra. Propose a structure, and show how it is consistent with the spectra. Show the fragmentations that give the prominent peaks at m/z...
-
Hexahelicene seems a poor candidate for optical activity because all its carbon atoms are sp2 hybrids and presumably flat. Nevertheless, hexahelicene has been synthesized and separated into...
-
An air stripping tower is being designed to treat carbon tetrachloride from 500 to 5 g/L at a temperature of 25 C with a stripping factor of 8. Determine the operating airtowater ratio
-
If the wind force has a magnitude of 65 N , with what tension must the worker pull in order to move the box at a constant velocity? Express your answer with the appropriate units
-
1. Write sonic anemometer, thermal (hot wire) anemometer, cup anemometer, or pitot tube after the statement that best describes its basic principle of operation. [4 points] a. Infers wind speed from...

Study smarter with the SolutionInn App


