Which of the following alkenes would you expect to be good Diels-Alder dienophile?s? (b) (a) H2C=CHCI %3H2H2C
Question:
Which of the following alkenes would you expect to be good Diels-Alder dienophile?s?
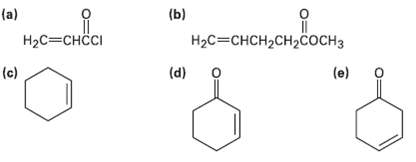
Transcribed Image Text:
(b) (a) H2C=CHCI Нас%3снсH2сH2Cоснз (d) (c) (e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Strategy Good dienophiles have an electronwithdrawing group conjugated with a double bond Good di...View the full answer

Answered By

Rohit anand
i have done btech from nit,hamirpur..i teach physics. i am teaching physics from last 2 year ..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Which of the following processes would you expect to be under control, and which would you expect not to be under control? Explain briefly why or why not. (a) Daily sales at each checkout line in a...
-
Which of the following nuclides would you expect to be radioactive: tungsten-184, polonium-206? Justify your choices. Ni, Cu, Ag, 47 108 Ag.
-
What is the square root of 3 to the square root of 2 power times the square root of 3 to the negative square root of 2 power?
-
How might a persistent global credit crisis affect the scale and scope of modern firms?
-
A balanced three-phase source furnishes power to the following three loads: Load 1: 6 kVA at 0.83 pf lagging Load 2: unknown Load 3: 8 kW at 0.7071 pf leading If the line current is 84.6 A rms, the...
-
Instead of the conventional unit operations, many B2C chemical products are manufactured using unconventional processing techniques such as etching, physical vapor deposition, and calendering....
-
Beaty Company has the following internal control procedures over cash receipts. Identify the internal control principle that is applicable to each procedure. (a) All over-the-counter receipts are...
-
A satellite is placed in a circular orbit to observe the surface of Mars from an altitude of 144 km. The equatorial radius of Mars is 3397 km. If the speed of the satellite is 3480 m/s, what is the...
-
Three-Month Project NOTE! Templates needed Ampersand, Inc., is a small business that operates in Somerset, VT The company is located at 732 Appalachian Way, Somerset, VT 05363. Its federal Employer...
-
Predict the product of the following Diels-Alderreaction: - C=C
-
Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate tos-cis? (a) (c) (b)
-
Brabant NV of the Netherlands is a wholesale distributor of Dutch cheeses that it sells throughout the European Community. Unfortunately, the company's profits have been declining, which has caused...
-
Write an assembly language program that asks the user to enter an integer dollar amount between 1 and 3,000. Your program should display the corresponding class description using the following table....
-
Plato in Meno discusses an innate trouble, on the off chance that not a conundrum, of doing investigate that can be summarized in the accompanying manner, "Assuming you realize what you're looking...
-
Driving in the streets of Boston is like driving through a maze. Most of the streets are narrow and winding, suitable more to horse-drawn carriages than to modern day vehicles. Also, almost all of...
-
Hart's primary interest is the role mass media plays in producing a sense of intimacy between us viewer/citizens and politicians. In many ways this is a false intimacy and has consequences, Hart...
-
Write an interactive C++ program that determines the status of a customer's loan application at a bank. The program should read in the customer's id number (e.g. C3182312), checking account balance,...
-
Find the inverse Laplace transform by using the partialfraction expansion method. \(\frac{s+1}{s^{2}(s+3)}\)
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
What are colligative properties?
-
Draw just the bonding -MO's for the cycloheptatrienyl cation. Draw the energy diagram to show the relative energies of all the MO's, and show which orbitals the electrons would occupy in the ground...
-
The proton NMR chemical shifts of the hydrogens in pyridine are shown. These are typical aromatic chemical shifts, except that the ortho protons (on the carbons bonded to nitrogen) are deshielded to...
-
Does the MO energy diagram of cyclooctatetraene (Figure 16-8) appear to be a particularly stable or unstable configuration? Explain. In Figure 16.8 nonbonding line cyclobutadiene cyclooctatetraene...
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


