Predict the products of the following reactions. Don?t worry about the size of the molecule; concentrate on
Question:
Predict the products of the following reactions. Don?t worry about the size of the molecule; concentrate on the functional groups.
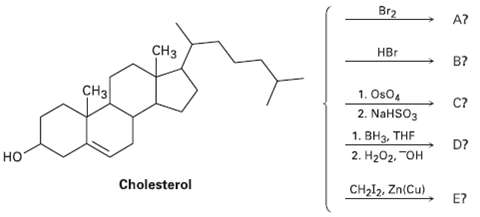
Transcribed Image Text:
Br2 A? CНз НBr B? CH3 1. OsO4 2. NaHSO3 C? 1. BH3, THE D? 2. Н20г. "он но Cholesterol CH2I2, Zn(Cu) E?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
HO HO H3G Br H3C Br Br2 H...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Austin and Anya Gould are a middle-aged couple with two childrenRusty, age 13, and Sam, age 11whom they adopted this year. They also bought a new home in the area to give the children a yard in which...
-
Download the full data set from the McGraw-Hill Connect website (only six data values are shown). (a) Use Excel to make a scatter plot, placing GDP per Capita on the X-axis and Birth Rate on the...
-
See Figure 11-19 and draw the cooling curves, including appropriate temperatures, expected for the following Al-Si alloys. (a) Al-4% Si; (b) Al-12.6% Si; (c) Al-25% Si; and (d) Al-65% Si.
-
Number of classes: 5 Data set: Regnal years of the monarchs of Great Britain Construct a frequency distribution and a frequency polygon for the data set using the indicated number of classes....
-
The Midwest Division of the Paibec Corporation manufactures subassemblies that are used in the corporations final products, Lynn Hardt of Midwests Profit Planning Department has been assigned the...
-
4. The slab of thickness H in the figure below is at temperature T, when the electric heat and convection conditions are suddenly applied. Set up the proper differential equation and conditions. hoo,...
-
Which one of the following items is remeasured using the current exchange rate under the temporal method? a. Accounts payable. b. Dividends declared. c. Additional paid-in capital. d. Amortization...
-
Which of the following alcohols could not be made selectively by hydroboration oxidation of an alkene? Explain. (a) ( , CH3CH2CH2CHCH3 (CH3)2HCICH)2 CH (c) (d) .CH " H.
-
The sex attractant of the common housefly is a hydrocarbon with the formula C23H46. On treatment with aqueous acidic KMnO4, two products are obtained, CH3 (CH2) 12CO2H and CH3 (CH2) 7CO2H. Propose a...
-
Suppose that federal marginal personal income tax rates will rise significantly over the next ten years. Explain the ways in which individuals at all levels of income can react over time, not just...
-
3.3 Find the fixed-point representation of the number 315.2342 in formats a. UQ16. b. UQ.16. c. UQ16.16.
-
VIGNETTE 2: THE PRINCESS BRIDE In the 1987 classic movie The Princess Bride, there is a conflict between Inigo Montoya and the "Man in Black," who, later to be learned, is Westley, the farmboy and...
-
Amount issued $ 290 million Offered Issued at a price of 98.75% plus accrued interest (proceeds to company 98.717%) through Citi and JPMorgan. Interest 9.25% per annum payable June 15 and December...
-
Assume that we have a fixed supply of a depletable resource to allocate between two periods. Assume further that the demand function is constant in the two periods, the marginal willingness to pay is...
-
What are the strengths perspective in social work as it applies to you ?
-
Explain how payments are calculated in an OIS.
-
Convert the numeral to a HinduArabic numeral. A94 12
-
Use MO theory to explain in detail why N 2 + and N 2 - have similar bond strengths and both are very different from neutral N 2 .
-
10.23lndicate which of the following balance the reactions are oxidation-reduction-reactions and which are not. For those involving oxidation-reduction, indicate which compound(s) are oxidized and...
-
How many moles of dichromate are required to oxidize one mole of ethanol to acetaldehyde? ethanol dichromate acetaldehyde
-
Give the product expected when each of the following alcohols reacts with pyridinium chlorochromate (PCC). HO--CH2CH2CH2 - OH
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
Miller Company ended its fiscal year on June 30, 2017. The company's adjusted trial balance as of the end of its fiscal year is shown below. MILLER COMPANY Adjusted Trial Balance June 30, 2017...
-
What depreciation method is used to write off property, plant and equipment? Regarding the triple bottom line, what information did the company disclose about environmental matters in the reports?...

Study smarter with the SolutionInn App


