In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example,
Question:
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl3 gives p-methylbenzaldehyde. Propose a mechanism.
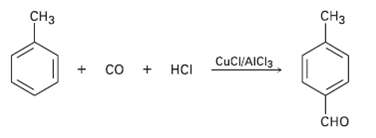
Transcribed Image Text:
CНз CHз со + нс CuC/AICI3 сно
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 36% (11 reviews)
1 HCI AICI 3 7C0 HCO AICI4 Carbon monox...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the aluminum chloride-catalyzed reaction of benzene with chlorine.
-
The benzene ring alters the reactivity of a neighboring group in the benzylic position much like a double bond alters the reactivity of groups in the allylic position. Benzylic cations, anions, and...
-
The Koch curve is a famous fractal introduced in 1906 by Swedish mathematician Niels Fabian Helge von Koch (1870-1924). To create a Koch curve, follow these steps: i. Draw a segment and divide it...
-
Fill in the blanks with the proper method names to deserialize an object. (Choose two.) A. writeObject in the first blank B. writeResolve in the first blank C. readObject in the first blank D....
-
Charlotte (age 40) is a surviving spouse and provides all of the support of her four minor children who live with her. She also maintains the household in which her parents live and furnished 60% of...
-
(a) In the analysis of a barrel of powder, the standard deviation of the sampling operation is 4% and the standard deviation of the analytical procedure is 3%. What is the overall standard...
-
Which event(s) in Exercise 6 can be considered unusual? Explain your reasoning. Data from Exercises 6 Basketball player Dwight Howard makes a free throw shot about 56% of the time. Find the...
-
Draw the shear and moment diagrams for thebeam 250 N/m -2 m- 2 m 500 N
-
Gilbert Canned Produce (GCP) packs and sells three varieties of canned produce: green beans; sweet peas; and tomatoes. The company is currently operating at 82 percent of capacity. Worried about the...
-
Explain how the business market differs from the consumer market for a product such as automobiles.
-
Propose a mechanism to account for the following reaction: C CH2CI AICI3
-
Treatment of p-tert-butyl phenol with a strong acid such as H2SO4 yields phenol and 2-methyipropene. Propose a mechanism.
-
a. Calculate the molar solubility of AgI in pure water. Ksp for AgI is 1.5 10-16. b. Calculate the molar solubility of AgI in 3.0 M NH3. The overall formation constant for Ag(NH3)2+ is 1.7 107. c....
-
South Africa Carbon Capture Systems (Pty) Ltd (SACCS' or 'the company') is a company that has a 31 December year-end. The company is NOT a small business corporation. The company is a South African...
-
Fox Manufacturing Company Income Statement for the Year Ended December 31, 2021 Sales revenue Less: Cost of goods sold Gross profits Less: Operating expenses General and administrative expenses...
-
1.A government bond fund buys a mix of 3-month treasury bills with a stated rate of 6% and a variety of treasury bonds with semiannual coupons. The firm is analyzing the implications if stated yield...
-
Why does Bernice have a decrease in arterial Po 2 (Pa co2 )? Why is her percent O 2 saturation decreased, and what are the implications for O 2 delivery to the tissues? Why are Bernices nail beds...
-
2. As a result of Cthulhu's body heat, the column of ocean water above him has roughly uniform temperature across all depths. According to the ideal gas law, the volume v of the gas bubble escaping...
-
Based on the ICs primary considerations for a combined portfolio, which simulated hedge fund strategy portfolio in Exhibit 1 creates the most suitable combined portfolio? A. Merger arbitrage B....
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
A Rankine steam cycle has been proposed to generate work from burning fuel. The temperature of the burning fuel is 1100 C, and cooling water is available at 15 C. The steam leaving the boiler is at...
-
Draw the structures for these compounds: (a) (Z)-Oct-3-en-2-one (b) 3-Ethylheptanal (c) 2, 4-Pentadienal (d) 3, 4-Dimethylbenzaldehyde (e) 1-Phenyl-1-propanone (f) 2, 2, 6, 6-Ttramythleyelohexanone
-
Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
-
(a) Is sodium hydroxide a strong enough base to completely remove a proton from the ?-carbon acetone; that is, does this equilibrium lie nearly completely to the right when sodium hydroxide is the...
-
Yi is collecting the Old Age Security pension benefit. Annually, Yi receives $5757 a year from OAS. In addition to OAS, Yi's annual income from her pension, CPP, and other sources totals $94250 a...
-
1. Write an equation that describes the information in the table: X Y -4 12 -2 3.5 2 -13.5 5 -26.25 2. Write an equation for the line in the graph below. Label the points on the graph that you used...
-
The inventory account is reduced by $36 to account for the returned defective items. what is the reason for this?

Study smarter with the SolutionInn App


