Reaction of cyclohexane with mercury (II) acetate in CH3OH rather than H2O, followed by treatment with NaBH4,
Question:
Reaction of cyclohexane with mercury (II) acetate in CH3OH rather than H2O, followed by treatment with NaBH4, yields cyclohexyl methyl ether rather than Cyclohexanol Suggest a mechanism.
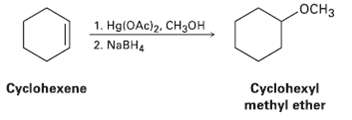
Transcribed Image Text:
OCH3 1. Hg(OAc)2, CH3OH 2. NaBH4 Cyclohexyl methyl ether Cyclohexene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
OAc HgOAc HgOAc Cy...View the full answer

Answered By

Pulkit Sahu
1. I have 3 years of tutoring experience in Chemistry subject.
2. 6 years of Industrial Experience in the field of oil and gas.
3. I like to solve problems in Chemistry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
SimmonsSmith reaction of cyclohexane with di iodomethane gives a single Cyclopropane product, but the analogous reaction of cyclohexane with 1, 1-diiodoethane gives (in low yield) a mixture of two...
-
Methyl acetate (CH3COOCH3) is formed by the reaction of acetic acid with methyl alcohol. If the methyl alcohol is labeled with oxygen-18, the oxygen-18 ends up in the methyl acetate: Do the C-OH bond...
-
A good Williamson synthesis of ethyl methyl ether would be What is wrong with the following proposed synthesis of ethyl methyl ether? First, ethanol is treated with acid to protonate the hydroxyl...
-
Wollongong Group Ltd, of New South Wales, Australia, acquired its factory building about 10 years ago. For several years the company has rented out a small annex attached to the rear of the building....
-
The U.S. Public Interest Research Group Education Fund, USPIRG, recently published a report titled The Campus Credit Card Trap: A Survey of College Students about Credit Card Marketing. You can find...
-
A micro laminate, Arall, is produced using five sheets of 0.4-mm-thick aluminum and four sheets of 0.2-mm-thick epoxy reinforced with unidirectionally aligned Kevlar fibers. The volume fraction of...
-
Using the frequency distribution constructed in Example 1, find the midpoint, relative frequency, and cumulative frequency of each class. Describe any patterns. Data from Example 1 The data set lists...
-
Multiple Choice Question 1. What is a subsidiarys functional currency? a. The parents reporting currency. b. The currency in which transactions are denominated. c. The currency in which the entity...
-
A 2m rod of mass 8 kg is supported by a (zero-friction) pin-joint at A. It is released from rest from the horizontal position. When the bar reaches the position as shown in the diagram,=60. Answer...
-
You are the president of XYZ Manufacturing company. You have recently learned about the following issue: A month ago, the company's general manager (GM) told one of the line managers to produce an...
-
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo- 2-methylcyclohexane. The analogous reaction of HBr...
-
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction: Co,CH3 C Hg(OAc)2 AcO-Hg
-
In the treatment of solid-liquid equilibria, the effects of pressure on melting points are neglected. (a) Draw a schematic of the Gibbs energy of liquid and solid phases versus pressure at constant...
-
3. The EU Directive requires "appropriate technical and organizational controls" to be in place to protect the confidentiality and integrity of personal data. How can an organization determine...
-
Work-life balance is a topic that has been discussed at length, especially about working mothers and single-parent households. There are definite challenges and also wonderful moments that face...
-
How does Miner's article "Ritual among the Nacirema" relate to ethnocentrism and cultural relativism, particularly concerning going to a GYM How does this play out in American culture? I want to...
-
What is the purpose of the electronic system in terms of every transaction in trust accounts? There is a word count below the text area. What is the purpose of the electronic system in terms of every...
-
When is an FBO required to carry hull insurance on their flight school planes? Discuss.
-
A national news organization developed the graphic shown in Figure 22 to illustrate the change in the highest marginal tax rate effective January 1, 2013. Why might this graph be considered...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
The ion CH 5 + can form under very special high-energy conditions in the vapor phase in a mass spectrometer. Propose a hybridization for the carbon atom and predict the geometry.
-
Give a curved- arrow mechanism for the rearrangement shown in {in Eq. 10.12.)
-
Complete the following reactions. If no reaction is tikety, explain why. (a) (b) CH,SH + NaOH -_, (1 equiv.) 25 C CH OH
-
Outline a synthesis of each ether using either alcohol dehydration or alkene addition, as appropriate. (a) 2-methoxy-2-methylbutane (b) dibutyl ether
-
3. Show that : -3t = y 3 sin(t) cos(t) + e is a solution to the differential equation y' + 3 y 10 sin(t)
-
The United States, Austrailia, Canada, and the United Kingdom cluster high on individualism but low on power distance on Hofstede\'s cutlutral graph. What does this mean to someone looking to market...
-
Mini-garden project Week one of the mini-garden project involved Sacha and her fellow pre-schoolers planting sunflower seeds into the outdoor garden bed. Each child in the activity has been assigned...

Study smarter with the SolutionInn App


