Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction: Co,CH3 C
Question:
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction:
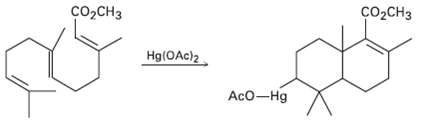
Transcribed Image Text:
Co,CH3 сооCнз Hg(OAc)2 AcO-Hg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
COCH3 COAC HgOAc ACO Aco COCH3 OAC COCH3 HOC 2 Hq AcO Hg Aco 3 COCH3 COOCH OAc The react...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Provide a mechanism for the following reaction and explain why it occurs faster than nitration of benzene. NO2
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Find the probability | (p)| 2d3 p of the particular momentum p for the ground-state hydrogen atom. (This is a nice exercise in three-dimensional Fourier transforms. To perform the angular...
-
A research study showed that adolescents who watched more than 4 hours of TV per day were more than five times as likely to start smoking as those who watched less than 2 hours a day. The researchers...
-
A laminate composed of 0.1-mm-thick aluminum sandwiched around a 2-cm-thick layer of polystyrene foam is produced as an insulation material. Calculate the thermal conductivity of the laminate...
-
The data set lists the out-of-pocket prescription medicine expenses (in dollars) for 30 U.S. adults in a recent year. Construct a frequency distribution that has seven classes. 200 239 155 252 384...
-
The following operating information reports the results of McKinney Company's production and sale of 16,000 air-conditioned motorcycle helmets last year. Based on early market forecasts, McKinney...
-
5. A three-stage, single acting compressor is required to compress 135m of free air per hour from 1bar to 64bar. Prove that with complete intercooling the stage pressure ratio should be equal to the...
-
On January 4, 2017, Macys Inc., faced with declining revenues and profits announced a series of actions it planned to implement to try to improve its operations. In its press release the company...
-
Reaction of cyclohexane with mercury (II) acetate in CH3OH rather than H2O, followed by treatment with NaBH4, yields cyclohexyl methyl ether rather than Cyclohexanol Suggest a mechanism. OCH3 1....
-
Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest a mechanism, using curved arrows to show electronmovement. CH2B Br2, H20 %3...
-
AB Ltd. issued 10,000 shares of 10 each. These shares were underwritten as follows: X: 3,000 shares; Y: 5,000 shares. The public applied for 7,000 shares, which included marked applications, as...
-
Lightner Inc. has the following inventory information available for the month of November: Date Description Number of Units Cost per Unit Nov 1 Beginning inventory 20 units $80 Nov. 5 Sales 15 units...
-
According to Lineberger, what are some of the social factors that contribute to health disparities?
-
What is an example of a successful organizational plan in criminal justice and an example of an unsuccessful organizational plan?
-
What strategy can be used to help students in Science or Social Studies? List two strategies and provide examples.
-
Coleen works for a newspaper in Hamilton, Ontario. Last year ( 2 0 2 2 ) , Coleen expects to earn $ 4 2 , 0 0 0 . How much would she have been required to contribute to CPP ? Ignore the employer...
-
The time-series graph in Figure 24 depicts the number of residents in the United States living in poverty. Why might this graph be considered misrepresentative? Approach Look for any characteristics...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
The bond angles increase steadily in the series PF 3 , PCl 3 , PBr 3 , and PI 3 . After consulting the data on atomic radii, provide an explanation for this observation.
-
Give the structure of the that would with mCPBA to give each of the following expoxides. (a) (b) . /A C CH2 H,C C-4 CH,
-
The chlorohydrins trans - 2 chlorocyclohexanol reacts rapidly in base to form an epoxide. The cis steroisomer, however, is relatively unreactive and does not give an epoxide. Explain why the two...
-
Explain the following facts with a mechanistic argument. (a) When the reaction mixture in part (a) is heated for times, l-iodobutane is also formed.
-
A) Translate the following C into RV641 assembly. Be sure to follow the calling (Refer to slide22 of lec6_postZoom.pdf) and comment each line of your assembly. Remember that long long is 64 bits in...
-
Given a Binary Tree, find the maximum sum path from a leaf to root. Input: 1 A Input: 10 -27 4 23 Output: Explanation Following the path 3-1, results in a sum of 4, which is the maximum path sum from...
-
On December 1 , 2 0 1 5 , your company pays $ 2 4 , 0 0 0 for rent on your store that covers the next 6 months. When you paid the cash, you debited Prepaid Rent.. The adjusting journal entry on...

Study smarter with the SolutionInn App


