Question: Repeat Problem 68 with a diatomic gas. B.
Repeat Problem 68 with a diatomic gas.
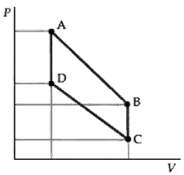
B.
Step by Step Solution
3.38 Rating (167 Votes )
There are 3 Steps involved in it
Proceed as in Problem 68 with C v 5 R and 14 The pressures volumes and temperatures are as sh... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
10-P-T-F-L (549).docx
120 KBs Word File


