(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain. CH3CH2CHCH3 2-Butanol...
Question:
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain.
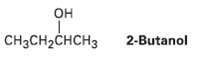
Transcribed Image Text:
Он CH3CH2CHCH3 2-Butanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
Since 2butanol is a secondary alcohol substitution can occur by e...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
On standing in dilute aqueous acid, compound A is smoothly converted to mevalonolactone. Suggest a reasonable mechanism for this reaction. What other organic product is also formed? CH3 CH3 CHj ...
-
On standing in dilute aqueous acid, compound A is smoothly converted to mevalonolactone.
-
(R)-2-Pentanol racemizes when placed in dilute sulfuric acid. Draw a mechanism that explains this stereochemical outcome, and draw an energy diagram of the process.
-
The probability that fewer than 35 people support theprivatization of Social Security A discrete random variable is given. Assume the probability of the random variable will be approximated using the...
-
What is knowledge management and why is it so important to today's employers?
-
What are some development strategies that many developers follow? Why do they follow such strategies?
-
Suppose there is a stock and a bond governed by the equations It is desired to construct a portfolio of these two securities that gives the maximum expected \(\log\) of return. However, although...
-
CEO was convinced by his employee, M. Ploy, that a coworker, A. Cused, had been stealing money from the company. At lunch that day in the company cafeteria, CEO discharges Cused from her employment,...
-
Develop a valuation model for the long-term corporate bond with a face value at maturity of $100,000, a maturity of 10 years, a coupon interest rate of 6%, and a market yield of 8%. The coupons are...
-
Design a Verilog code for a 3x3 array multiplier. Use structural Verilog description with full adders, half adders, and other gates. Submit your code, a testbench, and test results with a waveform....
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
The following is ABC, Inc.s, balance sheet (in thousands): Also, sales equal $ 500, cost of goods sold equals $ 360, interest payments equal $ 62, taxes equal $ 56, and net income equals $ 22. The...
-
Stacey owned all of the sock in small inc., a calendar year taxpayer, until april 30 when she sold it all to Andrew. Small made the following distributions during the year, april 1 $350,000 to stacey...
-
On December 31, 2022, Figg Company reported net sales revenue of $155,000, gain on the sale of equipment of $2,000, rent revenue of $4,000, and rent expense of $5,000. What would be reported as other...
-
In December, Mellie's Parfum's planned to sell 800 bottles of perfume but actually sold 900 bottles. Mellie Grant, the owner of the company, is reviewing the Flexible Budget Performance Report. The...
-
Discuss Zynga's revenue recognition policies, i.e., how Zynga recognizes its revenue, and whether its revenue recognition policy provides decision useful information in terms of relevance and...
-
Ahmed and Dubai Islamic Bank (DIB) have entered into a musharakah mutanaqisah contract to purchase a house. Initially, Ahmed owns 40% while DIB holds 60% ownership. The house costs AED 2,000,000,...
-
One consequence of defining the cumulant generating function in terms of the moment generating function is that the cumulant generating function will not exist any time the moment generating function...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
Write an expression for the equilibrium constant of each chemical equation. a. SbCl, (g) SbCl3(g) + Cl(g) b. 2 BINO (g) = 2NO(g) + Br(g) c. CH(g) + 2 HS(g) = CS(g) + 4 H(g) d. 2 CO(g) + O(g) = 2 CO(g)
-
Predict the products from crossed Claisen condensation of the following pairs of esters. Indicate which combinations are poor choices for crossed Claisen condensations. (a) (b) (c) (d) Ph -CH2 C-OCH,...
-
Show how crossed Claisen condensations could be used to prepare the following esters. (a) (b) (c) (d) Ph C-CH-C OCH,CH CH CH-C-OCH3 C-C-oCH Eto C CH-C OCH,CH Ph CH,CH,CH,
-
Predict the major products of the following crossed Claisen condensations. (a) (b) (c) NaOCH + Ph-C OCH NaOCH,CH CH,CH C CH CH,CHO C 0CH2CH NaOCH2CH
-
Company name is Walmart, Inc. here is the link https://www.sec.gov/ix?doc=/Archives/edgar/data/104169/000010416923000020/wmt-20230131.htm Overview 1. The financial statements for your company are...
-
Congress would like to increase tax revenues by 19 percent. Assume that the average taxpayer in the United States earns $54,000 and pays an average tax rate of 15 percent. Required: a. If the income...
-
Best Solutions is a retail merchandiser selling computer equipment. Best uses the gross method of accounting for inventory purchases and sales, a perpetual inventory system with LIFO inventory...

Study smarter with the SolutionInn App


