Show both the substitution and elimination products that are formed in these reactions: a) C CI +
Question:
Show both the substitution and elimination products that are formed in these reactions:
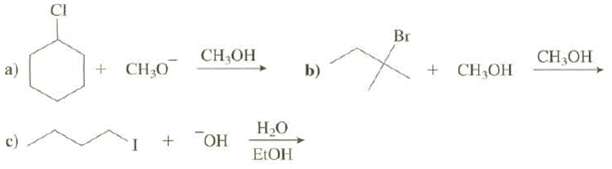
Transcribed Image Text:
a) C CI + CH₂0 CH₂OH + OH H₂O EtOH b) Br + CH₂OH CH3OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Elimination reactions compete with substitution reaction...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show both the substitution and elimination products that would be formed in these reactions: to , -CI a) Br CH,OH CH,O b) CI E:OH + CH;CH,OH
-
Give the substitution and elimination products you would expect from the following reactions. (a) 3-bromo-3-ethylpentane heated in methanol (b) 1-iodo-1-methylcyclopentane heated in ethanol (c)...
-
What substitution and elimination products (if any)might be obtained when each of the following alkyl halides is treated with sodium methoxide in methanol? (a) methyl iodide (b)...
-
Describe, in human terms, why delay and jitter are bad in real time (interactive) voice and video communications. Would these same problems apply to recorded voice and video stored and played back at...
-
The Launceston Wool Company designs and manufactures woollen coats, which are in high demand the cold Tasmanian winters. The company uses a process costing system to cost products because 1: produces...
-
Willkom Corporation bought 100 percent of Szabo, Inc., on January 1, 2011. On that date, Willkom's equipment (10-year life) has a book value of $300,000 but a fair value of $400,000. Szabo has...
-
Independent Nursing Consultants (INC) has the following data: If INC could streamline operations, cut operating costs, and raise net income to $\$ 300$, without affecting sales or the balance sheet...
-
1. Would you characterize television programming decisions as structured or unstructured? Explain. What type of decision-making condition would you consider this to be? Explain. 2. What criteria did...
-
Responding to activities that are regarded as computer crimes requires specific activities to be carried out from beginning to end. A. Name ANY TWO (2) positions for members included in an incident...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the rearranged carbocations that are expected from these carbocations: a) +CH CH3 b) CHCHCHCHCH c) CH3
-
When you whirl a can at the end of a string in a circular path, what is the direction of the force you exert on the can?
-
Two identical boxes A and B slide over the same rough floor. Box B stops over a distance twice as long as that needed for box A. Assuming a constant frictional force \(F\) acting in both cases,...
-
The engine of a moving spaceship suddenly stopped working. What will happen to the motion of the space ship?
-
Besides layoffs, what other options might the executives have tried to reduce costs?
-
The boiling point of water on top of a mountain reduced to \(80^{\circ} \mathrm{C}\) compared to \(100^{\circ} \mathrm{C}\) at the sea level. Is the boiling point of water intensive or extensive?
-
Plot the specified output by using the RK4 method. \(\left\{\begin{array}{ll}\dot{x}_{1}=x_{2} & x_{1}(0)=1 \\ \dot{x}_{2}=-\frac{1}{3} x_{1}-x_{1}\left|x_{1} ight|-x_{2}^{3}-1+\frac{2}{3} e^{-t /...
-
A heavy-oil stream at \(320 \mathrm{~K}\) is used in an absorber to remove dilute quantities of pollutant A from an air stream. The heavy oil is then recycled back to the process where A is stripped....
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Evaluate each using the values given. (-(-5-x)); use x = 6, and = = -2
-
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Draw structures corresponding to the following IUPAC names: (a) 2-Chloro-3, 3-dimethylhexane (b) 3, 3-Dichloro-2-methylhexane (c) 3-Bromo-3-ethylpentane (d) 1, 1-Dibromo-4-isopropylcyclohexane (e)...
-
The Morrit Corporation has $1,200,000 of debt outstanding, and it pays an interest rate of 8% annually. Morrit's annual sales are $6 million, its average tax rate is 25%, and its net profit margin on...
-
As an Investor, if I purchase a Corp Bond with a Coupon rate of 9%, and I am in the 30% tax bracket, effectively what rate will I be earning? Hint: If I purchased a Muni-Bond with an 8% Coupon rate,...
-
How can performance management systems be used to identify and nurture high-potential talent within the organization ?

Study smarter with the SolutionInn App


