Show both the substitution and elimination products that would be formed in these reactions: to , -CI
Question:
Show both the substitution and elimination products that would be formed in these reactions:
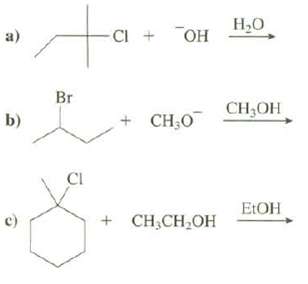
Transcribed Image Text:
to Н,О -CI он a) Br CH,OH CH,O b) CI E:OH + CH;CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
b substituti...View the full answer

Answered By

Simon kingori
I am a tier-one market researcher and content developer who has been in this field for the last six years. I’ve run the freelancing gamut; from market research, data mining and SEO/SMM to copywriting, Content Development, you name it, I’ve done it. I’m extremely motivated, organized and disciplined – you have to be to work from home. My experience in Freelancing is invaluable- but what makes me a cut above the rest is my passion to deliver quality results to all my clients- it’s important to note, I've never had a dissatisfied client. Backed by a Masters degree in Computer Science from MOI university, I have the required skill set and burning passion and desire to deliver the best results for my clients. This is the reason why I am a cut above the rest. Having taken a Bsc. in computer science and statistics, I deal with all round fields in the IT category. It is a field i enjoy working in as it is dynamic and new things present themselves every day for research and exploration.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show both the substitution and elimination products that are formed in these reactions: a) C CI + CH0 CHOH + OH HO EtOH b) Br + CHOH CH3OH
-
Show the aldol addition product that would be formed from each of the following compounds: a. b. c. d. CHCHCH-CHCH - - CH CHCH2CH CH CH3 ,
-
Give the substitution and elimination products for the following reactions, showing the configuration of each product: a. b. c. d. e. f. CH30 (S)-2-chlorohexane /E2conditions CH3OH (S)-2-chlorohexane...
-
The system shown consists of 3 cables. For example; cable C12 joins points 1 and 2. The coordinates of point 1 are (6.4, 0, 0) m, those of point 2 are (0, 9.5, -6.1) m, and those of point 3 are (0,...
-
How does the accounting for spoilage differ under the weighted average and FIFO methods of process costing?
-
The following transactions, adjusting entries, and closing entries were completed by Robinson Furniture Co. during a three-year period. All are related to the use of delivery equipment. The...
-
Why are most engineers likely to have experience with deterministic risk and not probabilistic risk?
-
The year 1987 featured extreme volatility on the stock market, including a loss of over 20 percent of the markets value on a single day. Figure 7.6 (a) shows the percent frequency histogram of the...
-
A project is expected to generate annual revenues of $129,700, with variable costs of $79,300, and fixed costs of $19,800. The annual depreciation is $4,600 and the tax rate is 21 percent. What is...
-
Financial information for American Eagle is presented in Appendix A at the end of the book. Required: 1. Calculate American Eagles percentage change in total assets and percentage change in net sales...
-
Show all of the steps in the mechanism and explain the stereo chemistry for this reaction: Br , Br + . + C-C CH3 . CH3 .
-
The reaction of 3-iodo-2, 2-dimethylbutane with ethanol gives three elimination products in addition to two substitution products as shown in the following equation. Show all the steps in the...
-
Study the numbered sentences in the following poorly written e-mail message. In teams or in a class discussion, identify specific sentence faults. To: Sierra.Maldonado@gmail.com From:...
-
Moser Company began business on March 1, 2012. At that time, it issued 25,000 shares of \(\$ 50\) par value, \(7 \%\) cumulative preferred stock and 80,000 shares of \(\$ 5\) par value common stock....
-
In a children's park, a boy uses a slide with inclination \(50^{\circ}\) with respect to the ground. Ignoring friction, calculate the magnitude of the acceleration of the boy.
-
Globalization affects businesses, but it also has a direct impact when a person a. uses technology to communicate with other gamers in Japan. b. has opportunities to study or work in countries around...
-
A ball is thrown vertically up with a speed \(10 \mathrm{~m} / \mathrm{s}\), caught by a boy leaning out of a window in 4 seconds. (a) Calculate the height of the window from the ground \((b)\) What...
-
The acceleration of a particle can be represented by the equation \(a(t)=t^{2}\). The particle begins accelerating from rest at \(t=0\). (a) What is its acceleration at \(t=2 \mathrm{~s}\) ? (b) What...
-
An open kettle contains \(50 \mathrm{kmol}\) of a dilute aqueous solution of methanol ( \(2 \mathrm{~mol} \%\) of methanol), at the bubble point, into which steam is continuously sparged. The...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
Sketch the graph of each line. y = 5.x -6- --- L m 2 11 x 9 STE t
-
Give the structures of the possible Claisen condensation products from the following reactions. Tell which, if any, you would expect to predominate in each case. (a) CH3CO2Et + CH3CH2CO2Et (b)...
-
Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate and ethyl 2-mcthylpropanoatc. Propose a mechanism for this...
-
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in...
-
Using the data above, fill out the missing information below: The average of the variable score is 45 The average of the variable submission is 1.2 The sample variance of the submission is 1.7 . The...
-
Financial information is presented here for two companies. (a) Fill in the missing amounts. Sales revenue Sales returns and allowances (a) Crane Company Bramble Company $94,000 (d) $ 5,400 87,000...
-
Hartford Research issues bonds dated January 1 that pay interest semiannually on June 30 and December 31. The bonds have a $40,000 par value and an annual contract rate of 10%, and they mature in 10...

Study smarter with the SolutionInn App


