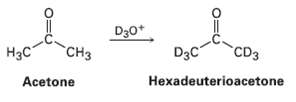
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone
Question:
Reaction of acetone with D3O+ yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid then propose a mechanism for this deuterium incorporation.
Transcribed Image Text:
D30* "CHз H3C D3C CD3 Acetone Hexadeuterioacetone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
24txA4 HC HC HC CH3 enol CH3 CH3 CH3 HC HCC HC ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mechanism involved in the reaction catalyzed by phosphoglyceromutase is known to involve a phosphory-lated enzyme intermediate. If 3-phosphoglycerate is radioactively labeled with 32P, the...
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
The hydration of an alkyne is not a reasonable preparative method for each of the following compounds. Explain why. (CH)C C CH
-
Kevin Steven opened a small tax-preparation service. Steven Tax Services trial balance at the end of its second year of operation is as follows. The following information is also available: a. Office...
-
Hill-O-Beans Coffee Company blends four component beans into three final blends of coffee: one is sold to luxury hotels, another to restaurants, and the third to supermarkets for store label brands....
-
Of the members of the Blue River Health Club, 43% have a lifetime membership and exercise regularly (three or more times a week). If 75% of the club members exercise regularly, find the probability...
-
Table 2.1 lists the history of 940 wafers in a semiconductor manufacturing process. Suppose that 1 wafer is selected at random. Let \(H\) denote the event that the wafer contains high levels of...
-
As Jacob walked back to his office after the weekly financial meeting, he reviewed his notes and saw that he had written down two operating income numbers: $110,000 and $50,000. He knew that the...
-
Details Email Exercise Purpose: To give students practice writing effective emails Instructions: Write a brief email to a friend or family member about your day. Be sure to include the following:...
-
There is no doubt that Johnson won the election, because an exit poll showed that she received 54% of the vote and the margin of error is only 3 percentage points. Decide whether the statement makes...
-
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Refer to Exercise 17.15. a. Find a 99% confidence interval for the hazard rate of the capacitors. b. Find a 99% confidence interval for the reliability of the capacitors at 3,000 hours. c. Find a 99%...
-
When you are trying to best manage your time, why should you resist the urge to multi-task?
-
25. 26. What are some factors that consumers should consider before selecting an insurance company? What is the role of a real estate broker? How can a homebuyer benefit from having a real estate...
-
Indian Railways proposes to implement a real time train tracking system for the newly introduced Vande Bharat trains. This system would have a real time tracking of the location of every train and...
-
Based on the law: NATIONAL LABOR RELATIONS ( 151 - 169) , what is the implication of this federal legislation in the education of United States?
-
With marketing and advertising we often "tell a story" so that you can draw in your audience. Think of many successful marketing campaigns - from Budweiser to Nike - and they often try to tell a...
-
The estimated equation for the linear U.S. corn demand curve is where \(p\) is the price in dollars per bushel and \(Q\) is the quantity demanded in billion bushels per year. \({ }^{5}\) What is the...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
What is a gas-evolution reaction? Give an example.
-
Give the relationships between the following pairs of structures. The possible relationships are: same compound, cis-trans isomers, constitutional (structural) isomers, not isomers (different...
-
Sulfur dioxide has a dipole moment of 1.60 D. Carbon dioxide has a dipole moment of zero, even though C-O bonds are more polar than S-O bonds. Explain this apparent contradiction.
-
For each of the following compounds, 1. Draw the Lewis structure. 2. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. 3....
-
In a company Christmas party, employees get one free spin of a wheel in an attempt to win a cash prize. The prizes and their probabilities are shown in the table. Prize ($) $10,000 $5,000 $500 $0...
-
What rate of return is currently being earned by such investment ? Were the corporate objectives met ? C Is the rate of return sufficient to cover the cost of capital ? What investment has been made...
-
what one piece of information is found on the statement of cash flows that is just as important as the bank balance (or more so) for the purpose of understanding the financial position of a company?

Study smarter with the SolutionInn App


