Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of
Question:
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it?
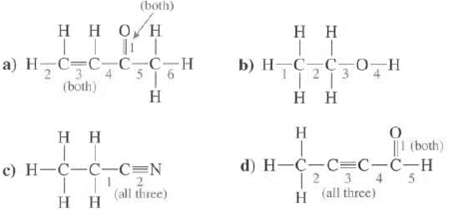
Transcribed Image Text:
(both) Η Η Ο Η TTT a) HC=C_C_CH Το (both) H Η Η 0 H-C-C-C=N 11 Η Η 2 (all three) Η Η || b) H_C_CO Η Η Η H |1 (both) d) H=C=C=C-C-H 1 2 3 4 H (all three)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
a 1 3 5 Csp2Osp2 and TTC2p02p 2 OCsp2H1s Csp2Csp2 and TTC2pCp2 4 OCsp2Csp2 Csp2...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
The general term that refers to the tendency of a parcel of air to either remain in place or change its initial position is ________. a. adiabatic b. conditional instability c. stasis d. stability
-
Explain why it may not be desirable for head office management to dictate transfer prices. Can you provide an example of a situation where this may be appropriate?
-
Sarah Wassim currently holds 400 shares of NutriFoods. The firm has 40,000 shares outstanding. The firm most recently had earnings avail able for common stockholders of US$80,000, and its stock has...
-
Describe normal flow, sub-flow, and alternate flow. How do they differ?
-
John R. lane (SSN 111-44-6666) lives at 1010 Ispen street, Yorba Linda, California 90102. John, a single taxpayer, age 66 provided 100% of his cousin's support. the cousin lives in Arizona. he wants...
-
1 . What is the starting point in doing a projected discounted cash flows analysis?A . Start with the projected increase in assets.B . Start with the required interest rate.C . Start with the sale...
-
Find v such that p, q, u in Prob. 21 and v are in equilibrium. Data from Prob. 21 Find the resultant in terms of components and its magnitude. p = [2, 3, 0], q = [0, 6, 1], u = [2, 0, -4]
-
What is the hybridization at all atoms, except hydrogen's in these compounds? a) CHNH, d) b) CH=CHCHC=N OH 6 H NH
-
What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H H H H H CH 4.6 c) HC-N-CH3 : : g) CHC-OH h) CHC-NHCH,
-
In Exercises 5 and 11, you described the relationship between height (in inches) and weight (in pounds) for Olympic track and field athletes. The scatterplot shows this relationship, along with two...
-
A cube that has a volume of \(1.00 \mathrm{~m}^{3}\) contains \(N\) distinguishable particles. Determine the probability that all the particles are in a smaller volume \(V\) of the cube if (a) \(N=1,...
-
John Walker Lindh is an American citizen who was captured by the Northern Alliance on the battlefield in Afghanistan in November 2001. Known as the American Taliban, Lindh attended a military...
-
\(\mathrm{A}\) box divided into identical compartments \(\mathrm{A}\) and \(\mathrm{B}\) contains two distinguishable particles in A and three distinguishable particles in \(B\). There are five...
-
You are on a spacecraft traveling away from the earth at 0.50c. A beacon on earth flashes exactly once per second. a. From the point of view of a person on earth, what is the time between one flash...
-
A box divided into identical compartments \(\mathrm{A}\) and \(\mathrm{B}\) contains 25 distinguishable particles in \(\mathrm{A}\) and 20 distinguishable particles in B. There are nine energy units...
-
Philosophers have, of late years, considered chiefly the tendency of affections, and have given little attention to the relation which they stand in to the cause which excites them. In common life,...
-
Trade credit from suppliers is a very costly source of funds when discounts are lost. Explain why many firms rely on this source of funds to finance their temporary working capital.
-
Prove that e x is equal to the sum of its Maclaurin series.
-
Draw structures for the following: (a) 2-Methyiheptane (b) 4-Ethyl-2, 2-dimethylhexane (c) 4-Ethyl-3, 4-dimethyloctane (d) 2, 4, 4-Trimethylheptane (e) 3, 3-Diethyl-2, 5-dimethylnonane (f)...
-
Draw a compound that: (a) Has only primary and tertiary carbons (b) Has no secondary or tertiary carbons (c) Has four secondary carbons
-
Draw a compound that: (a) Has nine primary hydrogens (b) Has only primary hydrogens
-
Select three services: one high in search attributes, one high in experience attributes, and one high in credence attributes. Specify what product characteristics make them easy or difficult for...
-
The New York City Council is ready to vote on two bills that authorize the construction of new roads in Manhattan and Brooklyn. If the two boroughs join forces, they can pass both bills, but neither...
-
Miss Israel has a chest tube in place for a spontaneous pneumothorax. You note that the water seal chamber is bubbling vigorously. What could cause this? What should you do? You are totaling intake...

Study smarter with the SolutionInn App


