What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H
Question:
What is the hybridization at all atoms, except hydrogen's, in these compounds?
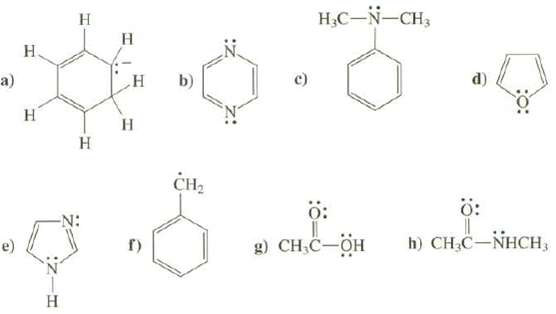
Transcribed Image Text:
a) H H H H H H H H CH₂ 4.6 c) H₂C-N-CH3 Ö: Ö: g) CH₂C-OH h) CH₂C-NHCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
H sp2 sp2 H H sp2 Sp2 H H sp2 H s...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
What is the hybridization at the indicated atoms in these compounds? a) CHCH=CH_NHCH3 12 3 c) CH,=CH0CH, 1 2 3 4 5 0: b) CHC0CH, 1 d) 2 1 NH
-
What is the hybridization of the indicated atom in each of the following molecules? a. b. c. d. e. f. CH CH CH CH CCH CH3CH2OH CH3CH NCH3 CH:OCH CH
-
In the proposal, the contractor estimated that 50 units of a specialty part are required. Each unit costs $300. There is a minimum buy requirement of 100 units. You contact the vendor and confirm the...
-
Explain how negotiation between the supplying and buying units may be used to set transfer prices. How does this relate to the general transfer pricing rule?
-
The Spurling Group is considering using magazine outlets to advertise their online Web site. The company has identified seven publishers. Each publisher breaks down its subscriber base into a number...
-
Supercritical, uniform flow of water occurs in a 5.0-m-wide, rectangular, horizontal channel. The flow has a depth of \(1.5 \mathrm{~m}\) and a flow rate of \(45.0 \mathrm{~m}^{3} / \mathrm{s}\). The...
-
Mountain Climbing Mountaineers often use a rope to lower themselves down the face of a cliff (this is called rappelling). They do this with their body nearly horizontal and their feet pushing against...
-
Let C' be the curve of intersection of the cylinder 25x + y = 25 and the plane x + y + z = 4. (a) Using t as the parameter where 0
-
A young woman named Seema (22) succumbed to injuries at a private hospital inGuwahati on 1st June, 2013 due to multiple organ failure as she had developedsevere health issues due to swallowing acid....
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it? (both) TTT a) HC=C_C_CH ...
-
Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c) H-C-C-CH=CH
-
In the application "Aggregating the Demand for Broadband Service" (based on Duffy-Deno, 2003), the demand function is Qs = 5.97p-0.563 for small firms and Ql = 8.77p-0.296 for larger ones. As the...
-
When locating an attorney in or outside your state, you can often do your searching online. Martindale-Hubbell has an online directory (http://www.martindale.com), as do most state bar associations....
-
Find the explicit solutions of the following stochastic differential equations: (a) Ornstein-Uhlenbeck process: \[d X_{t}=\mu X_{t} d t+\sigma d B_{t}\] (b) Mean reverting Ornstein-Uhlenbeck process:...
-
For turbulent flow of water in a pipe of diameter \(5 \mathrm{~cm}\) with \(R e=10^{5}\) estimate the magnitude of the length and velocity scales at which viscous dissipation becomes important.
-
If the per-phase line loss in a $60-\mathrm{km}$-long transmission line is not to exceed $60 \mathrm{~kW}$ while it is delivering 100 A per phase, compute the required conductor diameter, if the...
-
There is nothing magical about the eigenfunctions being orthogonal. This can be shown by integration by parts twice. Consider two eigenfunctions \(F_{n}\) and \(F_{m}\), both of which satisfy the...
-
Try to think of networks in different systems such as food webs, interconnected airports, social networks, etc. Use a web search to investigate which type of distribution the number of links attached...
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
Calculate lim 11 0 In n n
-
For each of the following compounds, draw an isomer that has the same functional groups. Each intersection of lines represents a carbon atom with the appropriate number of hydrogensattached. ( CH (a)...
-
Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH (a) H-H H CH3CH2CHCH2CH,CHCH3 (e) CH H CHCH2CH2CHCH2CH CH CH-H>H3 (d)
-
Name the five isomers of C6H14. Discuss.
-
According to Investopedia, the weekend effect is a phenomenon in financial markets in which stock returns on Mondays are often significantly lower than those of the immediately preceding Friday....
-
John and Jane Doe are both 40 years old with two children, ages 12 and 10. John is an assistant principal making $80,000 per year. He has been in the same school district for over 10 years, working...
-
What are the estimated share prices for all four companies in Q3-5? A table summarizing your findings is useful. The summary table here demonstrates our collective share prices for all four...

Study smarter with the SolutionInn App


