Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of
Question:
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it.
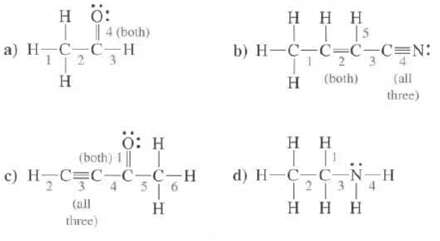
Transcribed Image Text:
Η Ο: a) H_C_C,H Η 4 (both) Ο: Η (beth)1] | 0 H_C_C_C=CH 4 16 Η Caill three) Η Η Η LIT b) H-C_C_C_C-N: H (both) Η Η Τ d) H=C=C, 1²1³1 Η Η Η 213 :NIH (all three) H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a 1 OCsp3 H1s b 1 OCsp3 Csp2 4 OCsp Nsp 2 OCsp3 Csp2 3 OCsp2 ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
The toroid of FIGURE P29.55 is a coil of wire wrapped around a doughnut-shaped ring (a torus). Toroidal magnetic fields are used to confine fusion plasmas. a. From symmetry, what must be the shape of...
-
lnkco manufactures printing inks. The company employs 150 people, of whom 120 are productt0 The ink market is extremely competitive and, with a 20 per cent market share, lnkco has the third largest...
-
A recent MBA graduate, Dara, has gained control over custodial accounts that her parents had established. Currently, her money is invested in four funds, but she has identified several other funds as...
-
Water flows in a 2-m-diameter finished concrete pipe so that it is completely full and the pressure is constant all along the pipe. If the slope is \(S_{0}=0.005\), determine the flowrate by using...
-
Clayton Industries has gathered the following information about the actual sales revenues and expenses for its pharmaceuticals segment for the most recent year (all data is in millions)....
-
Multiply. (10+5)(3/6+2)
-
a b d You have a new project for the construction of an 8km road in the Weija Municipality. The Client has requested that you submit a programme of works. Discuss three project planning techniques...
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
Suppose we surveyed 1000 adult Canadians about reform of the Canadian Senate. We found that 32% of people in Ontario supported abolishing the Senate of Canada, whereas the percentage in Quebec was...
-
What are the characteristics and advantages of the graphical user interface (GUI)?
-
Provide an example of centralized information system architecture.
-
What is the relationship of technology to the information system architecture?
-
What are some constraints in user interface design?
-
Comment on the statement that the essential purpose of the user interface is to enable the user and the system to exchange messages. Provide two examples.
-
What does the term "Management of Technology" refer to?
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Determine whether the series converges or diverges. 00 2n + 3n n=1 15 +
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
BRAND SELF COLLATERAL What is your brand name? What is your logo (YOU MUST DESIGN IT What is your brand slogan (the phrase that defines your brand and yourself) What are celebrity spokesperson is...
-
Amidst the burgeoning complexity of contemporary business landscapes, how do advanced organizations employ sophisticated design principles and ergonomic considerations to optimize physical workspaces...
-
JavaScript Assignment 1: Personal Web site This assignment is linked to your final project. Thus far, you have completed a total of 4 sections as part of the Final Project. You have created a basic...

Study smarter with the SolutionInn App


