Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b)
Question:
Show the products of these reactions:
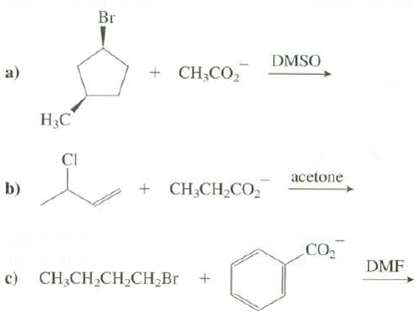
Transcribed Image Text:
Br DMSO + CH,CO, a) НС CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a H3C O...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these reactions of 1-propylcyclopentene with thesereagents: a) Br, CCl, b) Br2, H2O d) HBr f) [OsO,], t-BuOOH c) 1) BH3, THF; 2) H,O,, NaOH e) H2O, H,SO, g) 1) O; 2) (CHz)S
-
Show the products of these reactions: CH,OH a) (CH,CH),NH + CH,CH,Br ether b) CH;CH,NCH3 + CH,I CH3
-
Stock W, X and Y have expected returns of 9.0%, 16.1% and 11.3% respectively. Based on this and the attached information, what is the expected return of your portfolio? Stock W X Y Number of Shares...
-
Explain the impact of a decrease in the level of activity on: (a) Total fixed cost. (b) Fixed cost per unit of activity.
-
At point A on the demand curve shown, by what percentage will a 1 percent increase in the price of the product affect total expenditure on the product? 9 (units/week)
-
The random variable x represents the number of misprints in a page of a book. Determine whether the distribution is a probability distribution. If it is not a probability distribution, explain why. x...
-
Clyde had worked for many years as a chef executive of Red Industries INC and had also been a major shareholder. Clyde and the company had a falling out, and Clyde was terminated. Clyde and Red...
-
Find the derivative of the function. P(t) = 21+ t/7 P'(t) x
-
An assembly line consisting of three consecutive workstations produces two radio models: DiGi-l and DiGi-2. The following table provides the assembly times for the three workstations. The daily...
-
Because of the acidic conditions, this reaction proceeds by an SN1 mechanism. Which hydroxy group acts as the leaving group in the reaction? Show all the steps in the mechanism for this reaction: ,,
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
The Egret Company has a 40% combined Federal and state marginal tax rate. Egret's board estimates that, if its current president should die, the company would incur $200,000 in costs to find a...
-
How do you segment your market? Whos your PTA, your core customer? Who are your STAs?
-
What primary data will you collect?
-
What secondary sources can you use?
-
How do you learn about your customer?
-
What channels are available? Which channels are best? When will you add new channels?
-
In this exercise, we explore some of the factors predicting costs at American public universities using the data file pubcoll. Let \(T C=\) the real \((\$ 2008)\) total cost per student, \(F T U G=\)...
-
What is the purpose of the journal wizard?
-
20 British pounds is equal to $26.12. How many dollars is 12 pounds?
-
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2-hybridized, draw an orbital picture of...
-
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm. How do you account for this difference? -NH2 p-Toluidine U max = 235 nm) Benzene Amax = 204 nm)
-
What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, CH3CO2 ? Assume that inversion of configuration occurs, and show the stereo...
-
The sheet Inventory lists a grocery store's inventory for two months. Calculate the percent change for each item to two decimal places. If the formula gives an error, put "Initial Stock" in the cell....
-
A local manufacturer produces two different products on the same production line. Product A requires .5 hours of labor while product B requires .8 hours of labor. The total labor available in any one...
-
Hampton Company reports the following information for its recent calendar year. Income Statement Data Sales Expenses: Cost of goods sold Salaries expense Depreciation expense Net income Required:...

Study smarter with the SolutionInn App


