Question: Spectroscopic data for the indicators thymol blue (TB), semithymol blue (STB), and methylthymol blue (MTB) are shown in the table. A solution of TB, STB,
Spectroscopic data for the indicators thymol blue (TB), semithymol blue (STB), and methylthymol blue (MTB) are shown in the table. A solution of TB, STB, and MTB in a 1.000-cm cuvet had absorbances of 0.412 at 455 nm, 0.350 at 485 nm, and 0.632 at 545 nm. Modify the spreadsheet in Figure 18-5 to handle three simultaneous equations and find [TB], [STB], and [MTB] in the mixture.
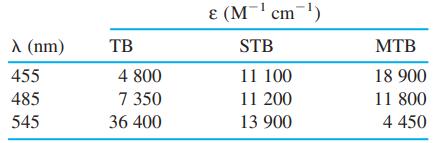
Figure 18-5
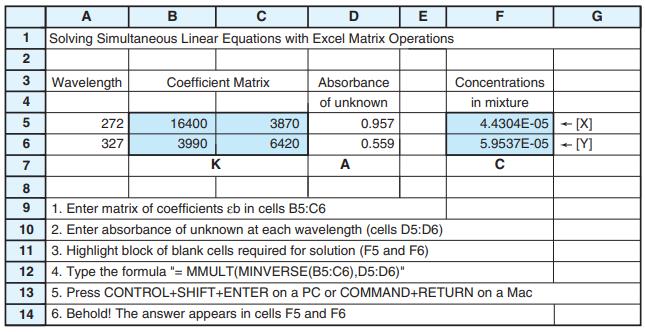
E (M' cm-) A (nm) TB STB MTB 455 4 800 11 100 18 900 485 7 350 11 200 11 800 545 36 400 13 900 4 450
Step by Step Solution
3.55 Rating (172 Votes )
There are 3 Steps involved in it
1 23456789 A B D E F Solving Simultaneous Linear Equat... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
878-E-C-E-E-C (2182).docx
120 KBs Word File


