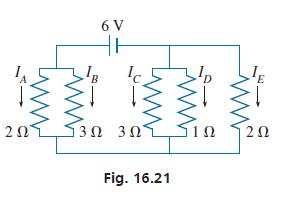
In applying Kirchhoffs laws to the circuit shown in Fig. 16.21, the following equations are found. Determine
Question:
In applying Kirchhoff’s laws to the circuit shown in Fig. 16.21, the following equations are found. Determine the indicated currents (in A).
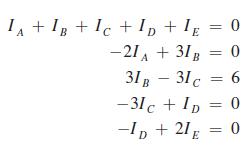
Transcribed Image Text:
IA +IB +IC+ID+1E = 0 -21A +31B = 0 31B - 31 c = 6 -31c + ID 0 C -ID + 21E = 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The circuit shown in Fig 1621 can be analyzed using Kirchhoffs laws Applying Kirchhoffs first law at ...View the full answer

Answered By

Mary Boke
I have teached the student upto class 12th as well as my fellow mates.I have a good command in engineering,maths and science.I scored 90+ marks in 10th and 12th in maths.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Basic Technical Mathematics
ISBN: 9780137529896
12th Edition
Authors: Allyn J. Washington, Richard Evans
Question Posted:
Students also viewed these Mathematics questions
-
Evaluate the line intigral I=[(x-8) ds aheve the semicircle of Tadies from A= (a, 0) to B=(-a,0) and for ahieh y20 is numing a
-
In applying Kirchhoffs laws to the circuit shown in Fig. 16.15, the following equations are found. Determine the indicated currents (in A). Fig. 16.15. Ic IA + B + IC = 0 = 6 21 A 51 B 51B - Ic = -3
-
Apply Kirchhoffs laws to the circuit shown in Figure 9.15 to determine the current that will be shown by the ammeters A 1 , A 2 and A 3 . 20 (A1 10 V (A2 202 5.0 V (A3) Figure 9.15: Kirchhoff's laws...
-
How to get a good answer using data and analysis tools in jmp 1 uses the other variables to predict "passion." passion would need to be aggregated into high/low first, and then you can use logistic...
-
Using the molecular orbital model to describe the bonding in CO, CO+, and CO2+, predict the bond orders and the relative bond lengths for these three species. How many unpaired electrons are present...
-
A student added 10 g (0.25 mol) of sodium hydroxide to 40 cm 3 of water to make a concentrated solution. All the sodium hydroxide dissolved. He measured the maximum temperature rise. He suggested...
-
0.0284 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04...
-
Digital, Inc., leased computer equipment from Young Leasing Company on January 2, 2012. The terms of the lease required annual payments of $4,141 for five years beginning on December 31, 2012. The...
-
In the context of solid-liquid extraction, how do particle size distribution, porosity, and surface area of the solid matrix impact extraction kinetics and overall efficiency? Provide examples of...
-
Solve the given systems of equations using the inverse of the coefficient matrix. 5A 7B = 62 6A + 5B = 6
-
Find the indicated matrices using a calculator. 6B 4A A = c = C 6-3 4 -5 B = -1 4 -7 2 -6 11 3 -9 D = 12 -6 79-6 -4 0 8
-
A New York Times article on grade inflation reported several findings related to a tendency for average grades to rise over the years and a tendency for the top-ranked institutions to give the...
-
Do you believe that companies can easily develop compensation that is both internally consistent and competitive within the market? What are some of the challenges to this goal? Provide an example of...
-
Why should Maersk's human resource management align with the organizations strategic plan
-
Compare and contrast recruitment and selection of internal versus external candidates using best practices from the Society for Human Resource Management (SHRM) website. Refer to the Research and...
-
Explain why it is important to deal with workplace violence and how it differsfrom sexual harassment.
-
Why is marketing research important to the development of a marketing strategy?Provide a specific example.
-
Evaluate the attempt by the FASB to separate stock options from stock appreciation rights that are payable in cash?
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
The regular price of a Phillips color TV is $539.62. During a sale, Hill TV is selling the TV for $439. Determine the percent decrease in the price of this TV. Where appropriate, round your answer to...
-
On January 1, Juan weighed 235 pounds and decided to diet and exercise. On June 30, Juan weighed 210 pounds. Determine the percent decrease in Juans weight from January 1 to June 30. Where...
-
The cost of a soccer ball to Soccer Line Sports Goods is $35. They sell the same soccer ball for $49. Determine the percent markup in the price of the soccer ball. Where appropriate, round your...
-
1. Discuss what Gauss's law is and its relationship with symmetry. Start by defining what flux is and how it is related to area. 2. Two equal and opposite charges of magnitude Q are located on the...
-
4.) Carbondioxide CO2 is a linear molecule with C-O bond length d=116.3 pm. The partial charges on the carbon and oxygen atoms are +28 and -8, respectively, with 8 = 0.73. The CO2 molecule is placed...
-
5 d) We define the path lengths L and L' between the landing and the two openings. Find the difference |L'-L| such that the condition from part c) is met! [note that there is no unique solution]. e)...

Study smarter with the SolutionInn App


