Calculate the pH of a buffer solution prepared by mixing 75 mL of 1.0 M lactic acid
Question:
Calculate the pH of a buffer solution prepared by mixing 75 mL of 1.0 M lactic acid (see Table 2.6) and 25 mL of 1.0 M sodium lactate.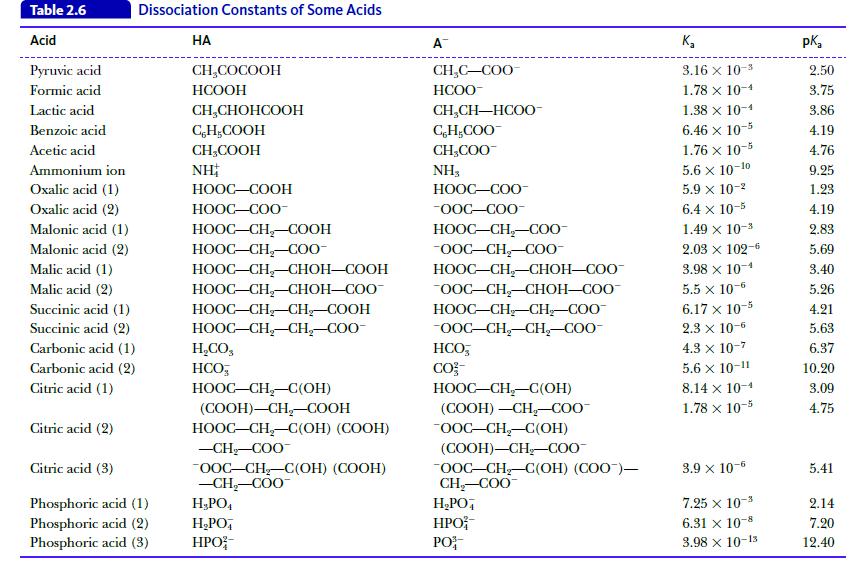
Transcribed Image Text:
Table 2.6 Acid Pyruvic acid Formic acid Lactic acid Benzoic acid Acetic acid Ammonium ion Oxalic acid (1) Oxalic acid (2) Malonic acid (1) Malonic acid (2) Malic acid (1) Malic acid (2) Succinic acid (1) Succinic acid (2) Carbonic acid (1) Carbonic acid (2) Citric acid (1) Citric acid (2) Dissociation Constants of Some Acids Citric acid (3) Phosphoric acid (1) Phosphoric acid (2) Phosphoric acid (3) HA CH₂COCOOH HCOOH CH₂CHOHCOOH CH₂COOH CH₂COOH NH HỌOC–COOH HỌOC—COO- HỌOC–CH,–COOH HOOC–CH,C00- HOOC–CH,–CHOH—COOH HOOC–CH,–CHOH–COO- HOOC–CH_CH, COOH HOOC–CH,CH,–COO- H,CO, HCO HOOC–CH,–C(OH) (COOH)-CH₂-COOH HOOC–CH,–C(OH) (COOH) -CH₂-COO OOC-CH₂-C(OH) (COOH) -CH₂-COO H₂PO4 H₂PO4 HPO A CH₂C-COO HCOO- CH,CH—HCOO- C,H,COO CH₂COO™ NH3 HỌỌC—COO- -OOC-COO- HOOC–CH,–C00- -OOC-CH₂-COO- HOOC–CH–CHOH–COO- OOC-CH₂-CHOH-COO™ HOOC–CH_CH_COO -OOC-CH₂-CH₂-COO- HCO, CO²- HOOC–CH–C(OH) (COOH) -CH₂-COO™ -OOC-CH₂-C(OH) (COOH)-CH₂-COO™ OOC-CH₂-C(OH) (COO)— CH₂-COO™ H₂PO4 HPO PO K₂ 3.16 x 10-3 1.78 x 10-1 1.38 x 10-1 6.46 x 10-5 1.76 x 10-5 5.6 x 10-10 5.9 x 10-² 6.4 x 10-5 1.49 × 10-3 2.03 × 102-6 3.98 x 10-4 5.5 x 10-6 6.17 x 10-5 2.3 x 10-6 4.3 x 10-7 5.6 X 10-11 8.14 x 10-1 1.78 x 10-5 3.9 x 10-6 7.25 x 10- 6.31 x 10-8 3.98 x 10-13 pka 2.50 3.75 3.86 4.19 4.76 9.25 1.23 4.19 2.83 5.69 3.40 5.26 4.21 5.63 6.37 10.20 3.09 4.75 5.41 2.14 7.20 12.40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
After mixing the buffer solution 100 mL contains 075 M ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Biochemistry
ISBN: 9781305961135
9th Edition
Authors: Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a buffer solution prepared by mixing 25 mL of 1.0 M lactic acid and 75 mL of 1.0 M sodium lactate.
-
(a) Calculate the pH of a buffer that is 0.12 M in lactic acid and 0.11 M in sodium lactate. (b) Calculate the pH of a buffer formed by mixing 85 mL of 0.13 M lactic acid with 95 mL of 0.15 M sodium...
-
The Kb for NH3 is 1.8 10-5 at 25C. Calculate the pH of a buffer solution made by mixing 65.1 mL of 0.142 M NH3 with 39.2 mL of 0.172 M NH4Cl at 25C. Assume that the volumes of the solutions are...
-
Problem A (20 points): Loco Farms Company sold 36,000 units of its only product and incurred a $18,672 loss (ignoring taxes) for the current year as shown here. During a planning session for year...
-
A monopoly is considering selling several units of a homogeneous product as a single package. A typical consumers demand for the product is Qd = 50 .25P, and the marginal cost of production is $120....
-
If a six-month Treasury bill is purchased for $0.9675 on a dollar (i.e., $96,750 for a $100,000 bill), what is the discount yield and the annual rate of interest? What will these yields be if the...
-
Which of the following is a characteristic of all plants? (a) seeds (b) pollen (c) swimming sperm (d) alternation of generations
-
Two bonds have the following terms: Bond A Principal ...$1,000 Coupon ...8% Maturity ...10 years Bond B Principal ...$1,000 Coupon ...7.6% Maturity ...10 years Bond B has an additional feature: It...
-
The Head of Tax has asked you to put together a one page TL;DR for the Tax File outlining the key impacts of the CARES Act on the company, specifically the payroll retention credit. She has also...
-
What is the relationship between pK a and the useful range of a buffer?
-
Calculate the pH of a buffer solution that contains 0.10 M acetic acid (Table 2.6) and 0.25 M sodium acetate. Table 2.6 Acid Pyruvic acid Formic acid Lactic acid Benzoic acid Acetic acid Dissociation...
-
What are some best practices for organizing transmittals and summaries?
-
Suppose Pepsicos stock has a beta of 0.57. If the risk-free rate is 3% and the expected return of the market portfolio is 8%, what is Pepsicos equity cost of capital?
-
Hubbard Industries is an all-equity firm whose shares have an expected return of 10%. Hubbard does a leveraged recapitalization, issuing debt and repurchasing stock, until its debt-equity ratio is...
-
A tarnished silver knife is placed in a beaker containing hot aqueous NaHCO 3 . A piece of Al foil is placed in the solution so that it touches the knife. The deposit of Ag 2 S disappears and the...
-
Should firms such as Aereo (see opening profile) be allowed to provide access to television broadcasts without their approval? Consider both sides of the argument to make a more convincing argument.
-
Given the difficulties in accurately predicting the future, is a business plan useful? Provide three reasons for writing one and three reasons for not preparing a plan. What is your conclusion and...
-
Write yes or no to indicate whether each of the following companies would typically use a job order costing system: 1. Soft drink producer 2. Jeans manufacturer 3. Submarine contracto 4. Office...
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
Why would a runner who has a 5-km race to run at 9 am be concerned about insulin?
-
Why do some people call GLUT4 the training glucose transporter?
-
How are insulin, GLUT4, obesity, and type II diabetes related?
-
Helinna Inc. manufactures custom jackets and uses job costing to calculate the cost of its jobs with direct labour hours as its manufacturing overhead allocation base. At the beginning of current...
-
Amarpreet Inc. has manufactured soccer balls for the World Cup. These balls are considered the best made in North America. Perfect specifications and durability are what makes them in demand....
-
Consider the binary search algorithm. Suppose A=(2,4,5,6,9,11,12,15,20). a) Suppose we are looking for the value 4. Which numbers will be compared with 4? How many total comparisons were performed?...

Study smarter with the SolutionInn App


