An ANOVA table is given: a.?Complete the ANOVA table.b.?How large was the sample?c.?Determine the standard error of
Question:
An ANOVA table is given: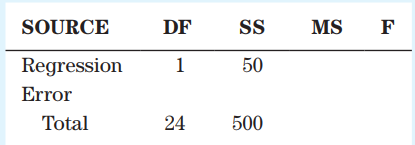
a.?Complete the ANOVA table.b.?How large was the sample?c.?Determine the standard error of estimate.d. Determine the coefficient of determination.
Transcribed Image Text:
SOURCE MS DF SS Regression 50 Error Total 24 500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
a b n 25 c s e 195652 44233 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Basic Statistics For Business & Economics
ISBN: 9781259268939
6th Canadian Edition
Authors: Douglas A. Lind, William G Marchal, Samuel A. Wathen, Carol Ann Waite
Question Posted:
Students also viewed these Mathematics questions
-
In a metallurgy experiment it is desired to test the effect of four factors and their interactions on the concentration (percent by weight) of a particular phosphorus compound in casting material....
-
The partially complete ANOVA table given here is for a two-factor factorial experiment: a. Give the number of levels for each factor. b. How many observations were collected for each factorlevel...
-
The following table shows a partially completed ANOVA table for a two-factor factorial experiment: a. Complete the ANOVA table. b. How many levels were used for each factor? How many treatments were...
-
We saw in Section 25.6 that DCC can be used to form a peptide bond. We explored the mechanism, and we saw that DCC activates the COOH moiety so that it readily undergoes nucleophilic acyl...
-
Show the locations of (a) alkali metals, (b) alkaline earth metals, (c) the halogens, and (d) the noble gases in the following outline of a periodic table. Also draw dividing lines between metals and...
-
Steam enters a turbine at 10 bar. The effluent pressure is 1 bar and the efficiency of the turbine is 80%. Determine the state of the turbine effluent (if pure liquid or vapor, find the temperature,...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Retail, LIFO Retail, and Inventory Shortage Late in 2007, Joan Seceda and four other investors took the chain of Becker Department Stores private, and the company has just completed its third year of...
-
What is operating leverage and how is it related to the cost structure of any organisation ?
-
On June 1, Cairns Corporation purchased goods from a foreign supplier at a price of 1,000,000 francs and will make payment in three months on September 1. On June 1, Cairns acquired an option to...
-
The chief of security for a shopping mall in Calgary, Alberta, was directed to study the problem of missing goods. He selected a sample of 100 boxes that had been tampered with and ascertained that...
-
A sample of 12 condominiums sold last week is given below. Can we conclude that as the number of bedrooms increases, the selling price (reported in $ thousands) also increases? a.?Compute the...
-
When certain expenditures result in tax credits that directly reduce taxes, the company will most likely record: A. a deferred tax asset. B. a deferred tax liability. C. no deferred tax asset or...
-
Given the combustion of butene, C 4 H 8 ( g ) + 6 O 2 ( g ) rightwards arrow 4 CO 2 ( g ) + 4 H 2 O ( g ) , determine the volume, to the nearest cubic metre, of CO 2 produced, at STP , when 9 9 kg of...
-
use nomenclature of complex ions. i ) What is the name for each of the following complex ions: a ) [ Fe ( H 2 O ) 3 ( NH 3 ) 2 NO ] 2 + b ) [ Ni ( CO ) 4 ] 2 + c ) [ Fe ( CN ) 6 ] 3 - d ) [ Co ( H 2...
-
Iron-59 is used to study iron metabolism in the spleen. Its half-life is 44 days. How many days would it take a 28.0 g sample of iron-59 to decay to 3.50 g? number of days: II
-
3 ) Provide proper IUPAC name for the molecule listed below. Detailed work should include i ) identification of functional groups, ii ) listed priorities of functional groups, 3 ) proper suffix and...
-
Unit 4 Hand - In Assignment # 2 This assignment covers sections 4 . 4 - 4 . 5 . Make sure you show all states for products and remember to balance equations. When using a formula, write down the...
-
Can a European option with a later exercise date be worth less than an identical European option with an earlier exercise date?
-
Which one of the following anhydrous chloride is not obtained on direct heating of its hydrated chloride? (A) BaCl2 (B) CaClz (C) MgCl2 (D) SrCl2
-
A random sample of the distances in miles 8 shoppers travel to their nearest supermarkets is shown. Test the claim at α = 0.10 that the standard deviation of the distance shoppers...
-
A researcher knew that before cell phones, a person made on average 2.8 calls per day. He believes that the number of calls made per day today is higher. He selects a random sample of 30 individuals...
-
How can the power of a test be increased?
-
Bank of Canada is the only controller of money supply. Suppose the Bank of Canada contracts the money supply. (a). Explain in words and draw graphs to show how the contractionary monetary policy?
-
What is your current taxable income? What would your taxable income be in the new job? (B) How much of your salary increase will be paid in payroll taxes?
-
= 2. Assume one equation for a good is P = 5000-300Q and a second equation for that good is P 2000+ 150Q. Assume Q is the quantity and P is price. Remember you must show all your math work. a....

Study smarter with the SolutionInn App


