Calculate the energy that must be supplied to raise the temperature of 5.0 kg of water from
Question:
Calculate the energy that must be supplied to raise the temperature of 5.0 kg of water from 20 °C to 100 °C.
You will need to use data from Table 19.3 to answer these questions.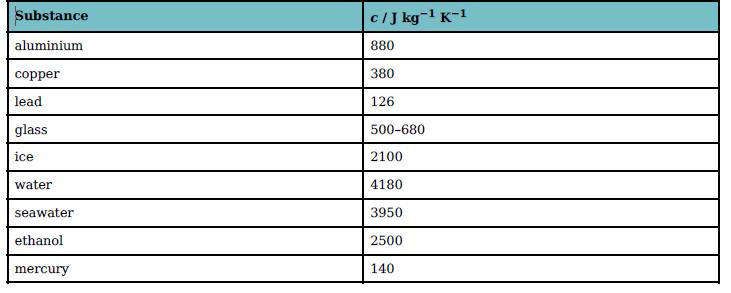
Transcribed Image Text:
Substance cIJ kg-1 K-1 aluminium 880 copper 380 lead 126 glass 500-680 ice 2100 water 4180 seawater 3950 ethanol 2500 mercury 140
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
we know the energy that must be supplied to raise th...View the full answer

Answered By

VAMSI TANGELLA
I have an experience of teaching about one year, i am good at solving questions and able to teach them in a way understandable by students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
The heat transfer necessary to raise the temperature of a constant-volume closed system is given by Q = U. Using the SL model, compare the heat necessary to raise the temperature by 10oC for such a...
-
Estimate the energy required to raise the temperature of 5 kg (11.0 lbm) of the following materials from 20C to 150C (68F to 300F): aluminum, brass, aluminum oxide (alumina), and polypropylene.
-
The heat required to raise the temperature of m (kg) of a liquid from T 1 to T 2 at constant pressure is in high school and in first-year college physics courses, the formula is usually given as (a)...
-
Discuss any of the established brands of the business concerned with woolworths in Australia
-
Jie & Partners purchased a computer priced at $949.99, financing it by paying $75.12 on the date of purchase, and signing a contract to pay equal monthly payments over the next 15 months. If the...
-
1. How did the laser cutter save Peerless Saw Company when it could not be justified on payback or ROI grounds? 2. Compare the decision Ted faces nowthe 1200-watt laser purchasewith the decision he...
-
How much energy is dissipated in the collision of Checkpoint 7.11? Data from Checkpoint 7.11 A 1000-kg compact car and a \(2000-\mathrm{kg}\) van, each traveling at \(25 \mathrm{~m} / \mathrm{s}\),...
-
On December 31, 2009, Durst Company's balance sheet showed the following balances related to its securities accounts: Durst's securities portfolio on December 31, 2009, was made up of the following...
-
Show work in terms of time lines or formulas ( No Excel) 4. A investment project generates the following incremental cash inflows over the next 5 years, C = $1.5 million, C = $1.3 million, C3 = $1...
-
Miller Heating is a small manufacturer of auxiliary heaters. The units sell for $300 each. In 2020, the company produced 1,000 units and sold 800 units. There was no beginning inventory. Below are...
-
When a thermocouple has one junction in melting ice and the other junction in boiling water it produces an e.m.f. of 63 V. a. What e.m.f. would be produced if the second junction was also placed in...
-
a. The first law of thermodynamics can be represented by the expression: U = q + W. State what is meant by all the symbols in this expression. b. Figure 19.18 shows a fixed mass of gas that undergoes...
-
What work should be done in order to squeeze all water from a horizontally located cylinder (Fig. 1.85) during the time t by means of a constant force acting on the piston? The volume of water in the...
-
Factor by grouping: xy +5y+10x + 50
-
1. Solve for x: 9 = 27
-
Question 3 Your company is in a legal dispute over a patent. You lost the first round in court, but have appealed. Despite the appeal, your attorneys tell you that you will most likely end up paying...
-
2 Solve the equation: m + 10m = 0 Answer: m =
-
Refer to the link below. Critical injuries, while unfortunate, are a real part of the workplace. Although infrequent, these situations require immediate intervention and investigation by competent...
-
Three point charges are placed on the corners of an equilateral triangle having sides of 0.150 m. What is the total electric force on the 2.50-μC charge? 2.50 HC 5.00 C -7.00 C
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Calculate K P at 600.K for the reaction N 2 O 4 (l) 2NO 2 (g) assuming that H o R is constant over the interval 298 725 K.
-
Consider the equilibrium CO(g) + H2O(g) ???? CO2(g) + H2(g). At 1150. K, the composition of the reaction mixture is a. Calculate K P and ÎG o R at 1150. K. b. Given the answer to part (a), use...
-
When 2,4-dibromo-3-methyltoluene is treated with bromine in the presence of iron (Fe), a compound with molecular formula C8H7Br3 is obtained. Identify the structure of this product.
-
Compute the student's GPA for the term. Round your answer to the nearest hundredth. A = 4 points, B = 3 points, C = 2 points, D = 1 point, F = 0 points Course Credits Grade MAT082 3 B ENG071 4 A...
-
6. Mrs. Rachel purchased a new car. The value, V, of the car can be modelled by the relation V = 49 100(0.945)", where n is the number of years since the car was purchased. a. What did Mrs. Rachel...
-
Simplify: 16q42 V Report your answer with a positive value of q.
MTEL Communication And Literacy Skills 01 Practice Test 2 1st Edition - ISBN: 1607872080 - Free Book

Study smarter with the SolutionInn App


