Use the binding energy graph (Figure 29.6) to suggest why fission is unlikely to occur with light
Question:
Use the binding energy graph (Figure 29.6) to suggest why fission is unlikely to occur with ‘light nuclei’ (A < 20) and why fusion is unlikely to occur for heavier nuclei (A > 40).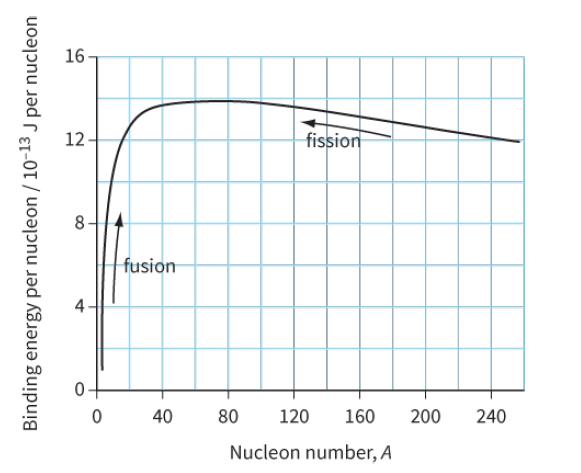
Transcribed Image Text:
16 12 fission fusion 40 80 120 160 200 240 Nucleon number, A -13 Binding energy per nucleon / 10- J per nucleon
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
Fission is unlikely to occur with light nuclei A 20 because the binding energy per nuc...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
Suggest a reason why the relaxation times of BC nuclei are typically much longer than those of IH nuclei.
-
Light nuclei can be split. For example, a deuteron, which is a proton-neutron combination, can split into a separate proton and separate neutron. Does such a process yield energy or cost energy?...
-
Use Fig. 30-1 to estimate the total binding energy of (a) 238/92U, and (b) 84/36Kr.
-
1. Consider the market for local rides (taxis, Uber Lyft, and so on), which is highly competitive. Suppose that the market is initially unregulated, but that the government imposes a binding price...
-
You are the accountant for Peel Credit Union. The lawyer for a member has sent a cheque for $7345.64 in full settlement of the members loan balance including interest at 6.25% for 11 months. How much...
-
The Giles Agency offers a 12% trade discount when providing advertising services of $1,000 or more to its customers. Audreys Antiques decides to purchase advertising services of $3,500 (not including...
-
Given that charge separation increases the electric potential energy of a system, what can you conclude about the criteria for keeping a system of positively and negatively charged particles in...
-
Four different companies, Sierra, Tango, Yankee, and Zulu, show the same balance sheet data at the beginning and end of a year. These data, exclusive of the amount of stockholders equity, are...
-
Question two (20mks) Building Societies Act is a well structured and balanced Organism that spells out different functions. Identify and describe the organization and management of building society...
-
Which measure shows more equality among countries around the world GNI calculated at market exchange rates GNI calculated at purchasing power parity? What explains the difference?
-
The graph of count rate against time for a sample containing indium-116 is shown. a. Use the graph to determine the half-life of the isotope. b. Calculate the decay constant. 160 120 80 40 10 20 30...
-
The proportions of different isotopes in rocks can be used to date the rocks. The half-life of uranium-238 is 4.9 10 9 years. A sample has 99.2% of the proportion of this isotope compared with newly...
-
From among the compounds chlorobenzene, o-dichlorobenzene, and p-dichlorobenzene, choose the one that (a) Gives the simplest 1H NMR spectrum (b) Gives the simplest 13C NMR spectrum (c) Has three...
-
Q1. A uniform rod of length L and mass M is attached at one end to a frictionless pivot and is free to rotate about the pivot in the vertical plane, as shown in Figure. The rod is released from rest...
-
Each of the following independent companies is missing numerical data. Required: Use your knowledge of the financial statement equations and their interrelationships to fill in the missing amounts....
-
what ways can active listening techniques be refined to foster deeper empathy and understanding during interpersonal exchanges?
-
A charge -1.9 nC is placed at (-3, 2, -3) m and another charge 6.4 nC is placed at (3, 3, -3) m. What is the electric field at (-3, 3, -2) m? (0.0.0) N/C
-
Find and simplify f(x+h). Simplify your answer. f(x)=-2x+7x-9 f(x+h) = X G
-
(a) How much heat does an engine with an efficiency of 33.3% absorb in order to deliver 1.00 kJ of work? (b) How much heat is exhausted by the engine?
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
Evaluate the commutator [ x, P 2 x ] by applying the operators to an arbitrary function f (x).
-
What is wrong with the following argument? We know that the functions n (x) = 2/a sin(n x/a) are eigenfunctions of the total energy operator for the particle in the infinitely deep box. We also know...
-
For linear operators A, B, and C, show that [A,BC] = [A,B]C + B[A,C].
-
What was the main problem with the older Legacy Windows NT networks?
-
Analyze and examine the steps involved in a typical forensics investigation methodology. Create a step table for yourself with the steps and the action taken in each step. Share your thoughts and...
-
1.1 Identify and Assess Potential and Actual Risks While being observed by your assessor, facilitate risk identification and assessment activity with program stakeholders (including project managers)...

Study smarter with the SolutionInn App


