a. What is the general trend in first ionisation energies across Period 3? b. Explain why aluminium
Question:
a. What is the general trend in first ionisation energies across Period 3?
b. Explain why aluminium has a lower first ionisation energy than magnesium.
c. Explain why sulfur has a lower first ionisation energy than phosphorus.
d. Look at Period 4 in the Periodic Table. The first ionisation energies of the p-block elements are given in the table below. Predict the missing value for the first ionisation energy of selenium.
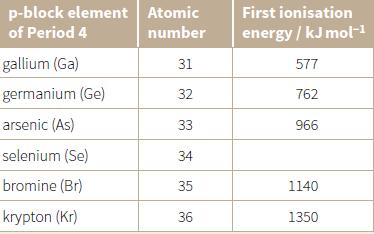
Transcribed Image Text:
p-block element Atomic First ionisation of Period 4 number energy / kJ mol-1 gallium (Ga) 31 577 germanium (Ge) 32 762 arsenic (As) 33 966 selenium (Se) 34 bromine (Br) 35 1140 krypton (Kr) 36 1350
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a The general trend in first ionisation energies across Period 3 from sodium to argon is that the fi...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
a. Look at Figure 11.3. i. What is the general trend in the melting points going down Group 2? ii. Which element breaks the trend? b. Explain why the atoms in Group 2, as in any other group, get...
-
What is the general relationship between mortgage rates and long-term government security rates? Explain how mortgage lenders can be affected by interest rate movements. Also explain how they can...
-
What is the general relationship among operating leverage, financial leverage, and the total leverage of the firm? Do these types of leverage complement one another? Why or why not?
-
A line charge density pL of length L lies parallel to an infinite sheet of surface charge density ps. How much work is required to rotate the line charge so that it is vertical?
-
Professor Stone complains that student teacher ratings depend on the grade the student receives. In other words, according to Professor Stone, a teacher who gives good grades gets good ratings, and a...
-
The activities described by the following table are given for the Howard Corporation in Kansas: a) Draw the appropriate AON PERT diagram for J. C. Howards management team. b) Find the critical path....
-
Between February 2020 and April 2020, the labor force participation rate for people ages 20 to 24 declined by 8.7 percentage points. The labor force participation rate for people ages 25 to 54...
-
Mini- Case: Nonaudit Services and Independence. Refer to the mini- case How Much Are Auditors Paid? shown on page C23 and respond to questions 1 and 2. 1. From a conceptual standpoint, how do the...
-
1. Consider two satellites S and S with periods of revolution 1hr. and 8 hr. respectively revolving around a planet in circular orbits. The ratio of angular velocity of satellite S to the angular...
-
Jayla just landed an internship with Acme Incorporated in the Payroll Department. She was excited because these internships usually turned into a full-time job after graduation. Jayla was hired by...
-
a i Describe how the atomic radius varies across Periods 2 and 3. ii. Explain this trend. b. i. Describe how the atomic radius varies down each group of the Periodic Table. ii. Explain this trend.
-
a. i. The Group 1 metal lithium reacts in a similar way to sodium. It reacts with oxygen, producing lithium oxide. Write the balanced symbol equation, including state symbols, for this reaction. ii....
-
Find the eigenvalues, eigenspaces, algebraic multiplicities and geometric multiplicities for the matrix below. It is possible to do all these computations by hand, and it would be instructive to do...
-
Manufacturing is related to a number of environmental impacts: acid rain, air pollution, microplastics in the environment, large landfills, and increased consumption of fossil fuels, among other...
-
How does the total economic profit compare to the total economic profit when the market had only one firm in it?
-
Truck drivers are in short supply these days, and that's putting a kink in the supply chain. What are the five factors might explain how the trucker shortage might affect aggregate supply? Will it...
-
Describe why we describe labor demand in the classical model as "technologically determined." Explain how preferences between (a) consumption goods purchased with money earned from working and (b)...
-
What types of evidence did opponents of ratification provide to support their argument that the United States was too large to govern? Explain briefly
-
Twin-Cities, Inc., purchased a building for $400,000. Straight-line depreciation was used for each of the first two years using the following assumptions: 25-year estimated useful life, with a...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
Based on your answer to Problem 17.67, propose a mechanism for the following transformation: Answer Problem 17.67 Heat CO2 heat
-
Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using...
-
Provide a systematic name for each of the following compounds. a. b. c. d. e. H. CH3 Br
-
What role do cultural factors, including norms around hierarchy, communication styles, and conflict management traditions, play in shaping conflict resolution approaches and influencing the...
-
(b) Consider the following three investment projects: A Cash-flow () Project Year 0 -900,000 12345 270,000 2 270,000 270,000 270,000 5 270,000 The relevant cost of capital is 10% B C Cash-flow ()...
-
2.1 The current Australian dollar exchange rate (Australian dollar per US dollar) is Et = 1.8; the annualized interest rate on three year bonds is 2% in Australia compared to a 1% annualized interest...

Study smarter with the SolutionInn App


