a. Write the rate equation for the acid-catalysed reaction of iodine with propanone. b. Use your rate
Question:
a. Write the rate equation for the acid-catalysed reaction of iodine with propanone.
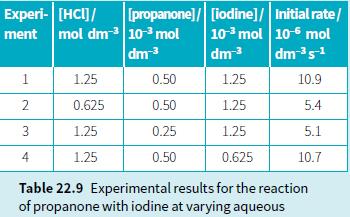
b. Use your rate equation and the information in Table 22.9 (experiment 1) to calculate a value for the rate constant for this reaction.
Transcribed Image Text:
Experi- [HCI]/ ment mol dm 10- mol dm 3 [propanone]/ [iodine]/ Initial rate/ 103 mol 10-6 mol dm3 dms1 1 1.25 0.50 1.25 10.9 2 0.625 0.50 1.25 5.4 1.25 0.25 1.25 5.1 4 1.25 0.50 0.625 10.7 Table 22.9 Experimental results for the reaction of propanone with iodine at varying aqueous 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
A The reaction is T he reaction is first ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
The rate equation for the reaction between iodine and propanone is: rate = k[CH 3 COCH 3 ] [H + ] [I 2 ] 0 a. State the order of reaction with respect to iodine. b. State the overall order of...
-
When you calculated A in the rate equation for the reaction of kMnO 4 solution and H 2 C 2 O 4 solution. you assumed k had the same value under the conditions of determinations 1, 2, and 3. (a) What...
-
Write the balanced equation for the reaction of sulfurous acid with dichromate ion.
-
Jacky Ma Ltd sells a single product called Alibaba. During 2020, 10,000 units were produced and 9,500 units were sold. There was no work-in-process inventory on 31 December 2020, that is the...
-
(a) Compute the coefficient of variation. (b) Compute a 75% Chebyshev interval around the sample mean.
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
At present, 10-year Treasury bonds are yielding 5 percent, while a 10-year corporate bond is yielding 7 percent. If the liquidity-risk premium on the corporate bond is 0.5 percent, what is the...
-
1. Jeanie acquires an apartment building in 2003 for $260,000 and sells it for $500,000 in 2014. At the time of sale there is $78,000 of accumulated straight-line depreciation on the apartment...
-
123456789 10 What is the need of MIS? What are the objectives of MRP? JIT introduced in which country? Advantages of TQM. Discuss about six sigma. Define various levels of CMM. What is mean by supply...
-
Dr. David Kipper filed a defamation lawsuit in a New York trial court against NYP Holdings Co. (NYP), the owner and publisher of the New York Post. Kipper based his case on an article in the December...
-
An acidified solution of hydrogen peroxide reacts with iodide ions. H 2 O 2 (aq) + 2H + (aq) + 2I (aq) 2H 2 O(l) + I 2 (aq) The rate equation for this reaction is rate = [H 2 O 2 ] [I ] The...
-
a. State the order of reaction for the decomposition of nitrogen(V) oxide. b. Use the data for 3.00 mol dm 3 N 2 O 5 in Table 22.8 to calculate a value for the rate constant for this decomposition....
-
This erionite was formed as an alteration product of what volcanic material?
-
When working in teams, students and employees often don't allow for adequate preparation time before beginning the team's tasks. Instead of immediately jumping to the task at hand, it is recommended...
-
Eight years ago, Natalia purchased an 8.25% $1,000 bond for 107 1/2 plus brokerage fees totaling $28. Natalia received interest checks semiannually Immediately after receiving her 16th interest...
-
Please have an elevator pitch for the case below and answer the following questions: (Please try to be concise and write a short answer for each question.) 1) Does Biolite illustrate the principles...
-
For the data below, which represents a sample with n = 9, answer the questions. Round to 4 decimal places where possible. x 14.3 14.6 14.5 13.7 11.7 8.5 28.9 4.4 16.3 Find the mean: Find the median:...
-
What a response to a peer that states In financial distress, a company or individual cannot generate enough income or revenue to meet its financial obligations. It can be caused by various factors,...
-
A camera lens (n = 1.50) is coated with a thin film of magnesium fluoride (n = 1.38) of thickness 90.0 nm. What wavelength in the visible spectrum is most strongly transmitted through the film?
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
The Youngs modulus (see P2.40) of muscle fiber is approximately 2.80 10 7 Pa. A muscle fiber 3.25 cm in length and 0.125 cm in diameter is suspended with a mass M hanging at its end. Calculate the...
-
DNA can be modeled as an elastic rod which can be twisted or bent. Suppose a DNA molecule of length L is bent such that it lies on the arc of a circle of radius R c . The reversible work involved in...
-
A 1.75 mole sample of an ideal gas is compressed isothermally from 62.0 L to 19.0 L using a constant external pressure of 2.80 atm. Calculate q, w, U, and H.
-
The following three identical units of Item LO3V are purchased during April: April 2 April 15 April 20 Total Average cost per unit Item Beta Units Cost Purchase 1 $191 Purchase 1 194 Purchase 1 197 3...
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
Watts and Lyon are forming a partnership. Watts invests $42,000 and Lyon invests $63,000. The partners agree that Watts will work one-third of the total time devoted to the partnership and Lyon will...

Study smarter with the SolutionInn App


