The table lists the boiling points of some Group 15 hydrides. a. Explain the trend in the
Question:
The table lists the boiling points of some Group 15 hydrides.
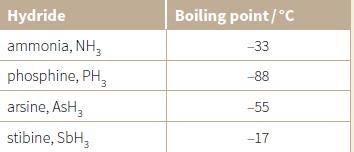
a. Explain the trend in the boiling points from phosphine to stibine.
b. Explain why the boiling point of ammonia does not follow this trend.
Transcribed Image Text:
Hydride Boiling point/°C ammonia, NH, -33 phosphine, PH, -88 arsine, AsH, -55 stibine, SbH, -17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Solution agenerally melting and boiling points of compounds increases down the groupfrom phophine t...View the full answer

Answered By

Monish Ramesh
Tuttored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
I teach Chemistry since 2 years, the best experience that I would like to share is that irrespective of basics that an individual has, need to help the student in understanding the core concepts in clarity. In that way, I am able to push my students in understanding the basic logical structure of subject and excel slowly. I prefer and pump the concepts slowly rather than going in a super sonic speed. For any student (irrespective of class) acquiring interest in the subject matters rather than learning like a machine.
Read
understand
understood
register
recollect
apply
innovate
invent
these are the fundamental steps to progress a learning curve in the subject.
For the last one+ year, I have been teaching “How to teach online”
Why? Because many good tutors out there do not know of tech tools that can make online teaching more efficient and engaging.
Other than than that, I have done some sessions on “Problem Solving and Decision Making” for folks in the IT industry. Basically, I enjoy sharing experiences
The back and forth and the light-bulb moments when students see something they hadn't before are a great stimulation.
If you want an ever-varying career, then teaching is an option. BUT not knowing where you are I would say have a talk with teachers in your country.
Tutoring is something else. Usually one on one, but I prefer two students at a time, so they can spark one another off and teach one another.
BUT don't think that doing it for free or for very little is doing them a favour
I’ve had the fortunate opportunity to give some guest lectures on software build infrastructure. Also I’ve given some training on design patterns and various short sessions on various topics.
I quite enjoyed doing those actually, so far that I did consider it as a career more than once.
If iam interested in learning a new language, I will teach someone in my mother tongue and learn theirs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
The table shows the atomic number and boiling points of some noble gases. a. Use ideas about forces between atoms to explain this trend in boiling points. b. Xenon forms a number of covalently bonded...
-
Explain why a. H2O has a higher boiling point than CH3OH(65oC). b. H2O has a higher boiling point than NH3(- 33oC). c. H2O has a higher boiling point than HF (20C).
-
This table indicates the freezing and boiling points of several molecules: Molecule Freezing Point (C) Boiling Point (C) Water 0 100 Carbon tetrachloride (CCl4) - 23 77 Methane (CH4) - 182 - 164...
-
Find all values of 0, if 0 is in the interval [0, 360) and has the given function value. cot 0= -1 0= (Type your answer in degrees. Use a comma to separate answers as needed.)
-
Use your calculator to evaluate these expressions. Express the final answer in proper scientific notation. a. 456 (7.4 108) =? b. (3.02 105) ( (9.04 1015) =? c. 0.0044 0.000833 = ?
-
The accounts in the ledger of Time Is Money Delivery Service contain the following balances on July 31, 2015. Accounts Receivable ......... $10,642 Prepaid Insurance ............ $ 1,968 Accounts...
-
Two parallel-plate capacitors 1 and 2 are identical except that capacitor 1 has charge \(+q\) on one plate and \(-q\) on the other, and capacitor 2 has charge \(+2 q\) on one plate and \(-2 q\) on...
-
Donaldson Corporation is a manufacturer of computer accessories. It uses absorption costing based on standard costs and reports the following data for 2014: There are no price spending, or efficiency...
-
8.The following data is available: Country Y currency Dollar Country X currency Peso Country Y interest rate 1% per year Country X interest rate 3% per year Country X expected inflation rate 2% per...
-
A store has determined that the number of Blu-ray movies sold monthly is approximately n ( x ) = 6750(0.929 x ) movies where x is the average price in dollars. (a) Write the function for the model...
-
Describe the changes that occur in the closeness and motion of the particles when: a. A solid changes to a liquid b. A liquid changes to a gas.
-
Bromine, Br 2 , and iodine monochloride, ICl, have the same number of electrons. But the boiling point of iodine monochloride is nearly 40 C higher than the boiling point of bromine. Explain this...
-
In a sample of 2016 U.S. adults, 242 said Richard Nixon was the worst president since World War II. Three U.S. adults are selected at random without replacement. (a) Find the probability that all...
-
Turning first to our current development activity. As you can see on Slide 11, we completed $740 million in new developments for the year which meets our initial underwriting and delivers very...
-
Provide at least one benefit a taxpayer can receive related to their investment income.
-
On December 31, 2019, McCullouch Enterprises recorded a deferred tax asset of $160,000 due to the potential tax benefits of an operating loss carry forward. McCullouch's enacted tax rates are as...
-
Assume that MSQM Blockchain Incorporated provided services to customers during 2021 at a total value of $900,000, of which $130,000 was collected in cash during 2021; the balance of 770,000 will be...
-
A taxpayer buys a tract of land with a small house on it. The house sits vacant for several months but finally, on December 1, Year One, they rent the property to Mr. Lance for one year. The rental...
-
How many ethers have the molecular formula C5H12O? Give the structural formula and systematic name for each. What are their common names?
-
Explain the operation of the dividends received deduction.
-
Draw all constitutional isomers with molecular formula C 5 H 10 that possess one p bond.
-
Rank the following anions in terms of increasing basicity:
-
Identify the alkene that would yield the following products via ozonolysis: a. b. c. d.
-
You purchased shares of Disney ten years ago that cost $1000 in total. Today the shares are worth $2800. What is the annualized rate of return on the shares of Disney? Note: This is an investment...
-
1/GroRight Inc. is building a new warehouse facility at a cost of $12.4 million. The CEO of the firm is quite conservative and will only go ahead with this project once the company has sufficient...
-
Royal Industries has for many years enjoyed moderate but stable growth in sales and earnings. In recent years, it has faced a stiff competition in its plastic product line and, consequently, its...

Study smarter with the SolutionInn App


