Photosynthesis to produce sucrose C 12 H 22 O 11 can be considered to occur by the
Question:
Photosynthesis to produce sucrose C12H22O11 can be considered to occur by the following reaction
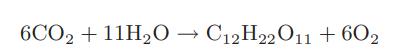
How much Gibbs free energy at 25°C must be obtained from sunlight for each mole of sucrose produced?
Transcribed Image Text:
6CO2 +11H₂O → C12H22011 +602
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine the amount of Gibbs free energy obtained from sunlight for each mole of sucrose produced in photosynthesis we need to calculate the Gibbs ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The free-energy change (G') for the oxidation of the cytochrome aa3 complex by molecular oxygen is 102.3 kJ = 224.5 kcal for each mole of electron pairs transferred. What is the maximum number of...
-
(a)At 0 o C, liquid water and ice are in equilibrium at standard pressure, so they have the SAME molar Gibbs energy. When raise from 1.0 bar to 100 bar, how much Gibbs free energy change in the...
-
Graph the function y = (x + |x|). At what values of x does the derivative exist and what is the value of the derivative when it does exist?
-
Identify some possible causal factors for the following support departments: a. Cafeteria b. Custodial services c. Laundry d. Receiving, shipping, and stores e. Maintenance f. Personnel g. Accounting...
-
The Bayside Art Gallery is considering installing a video camera security system to reduce its insurance premiums. A diagram of the eight display rooms that Bayside uses for exhibitions is shown in...
-
Find the = 0.05 critical value for the chi-square distribution with 18 degrees of freedom.
-
Assume that Revolights was interested in working with strategic partners in order to enhance the firms success. What types of companies might Revolights partner with to become more successful?
-
what is the most mean-variance efficient portfolio? is the one with reward to variability ratio most closely to 1? or the maximum one?
-
Another way to reduce the amount of nitric oxide is exhaust gasses is to inject ammonia resulting in the following reaction Determine the equilibrium conversion of nitric oxide at 1000C and 1 bar by...
-
An equimolar mixture of ethylene and hydrogen chloride react in the gas phase at 150C and 10 bar to form ethyl chloride. Determine the equilibrium composition at these conditions.
-
Explain two approaches to computing depreciation for a fractional period in the year in which an asset is purchased. (Neither of your approaches should require the computation of depreciation to the...
-
On January 1 , 2 0 2 3 , Fisher Corporation purchased 4 0 percent ( 7 2 , 0 0 0 shares ) of the common stock of Bowden, Incorporated, for $ 9 9 2 , 0 0 0 in cash and began to use the equity method...
-
The income statement and balance sheet of Sheng Siong Group Ltd ("Sheng Siong"), one of Singapore's largest retailers with 67 stores located across the island. over the last three years are provided...
-
Golf Academy, Incorporated, provides private golf lessons. Its unadjusted trial balance at December 3 1 , 2 0 2 1 , follows, along with information about selected accounts. Account Names Debit...
-
Identify the major purpose of cost accounting. Know the two basic types of cost accounting systems. Know the important features of a job order cost system. Know the steps in the flow of costs. Know...
-
A manufacturing company reports the following for the period: Inventories Beginning Ending Raw materials $ 1 8 , 1 2 0 $ 1 2 , 1 0 0 Work in process 9 , 5 0 0 1 1 , 3 0 0 Finished goods 1 2 , 5 6 0...
-
Well-Bred Service Company earned $50,000,000 during 2012 and paid $20,000,000 in dividends to the holders of its 40 million shares. If the current market price of Well-Breds stock is $31.25,...
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Investigate whether the equilibrium can be maintained. The uniform block has a mass of 500 kg, and the coefficient of static friction is s = 0.3 200 mm A 600 mm 800 mm
-
The homogenous semicylinder has a mass of 20 kg and mass center at G. If force P is applied at the edge, and r = 300 mm, determine the angle at which the semicylinder is on the verge of slipping....
-
The uniform crate has a mass of 150 kg. If the coefficient of static friction between the crate and the floor is s = 0.2, determine whether the 85-kg man can move the crate. The coefficient of...
-
Define uncovered interest rate parity (UIP). Derive the equations of UIP in both levels and logs. (b) Let the spot rate between the UK and Canada be 3.5 CAD/GBP, and the Canadian 6 month (annualized)...
-
Explore the concept of the "antihero" in contemporary fiction. How does this character archetype differ from traditional heroes, and what does its popularity suggest about changing societal attitudes...
-
A company's fixed operating costs are $670,000, its variable costs are $3.15 per unit, and the product's sales price is $5.50. What is the company's break-even point; that is, at what unit sales...

Study smarter with the SolutionInn App


