The reaction ClO 3 + 3H 2 SO 4 Cl + 3SO 4 = +
Question:
The reaction ClO3− + 3H2SO4 → Cl− + 3SO4= + 6H+ in 0.2N H2SO4 was studied in 1932 by Nixon and Krauskopf, who reported the data in Table 6.P2. Find a rate expression that is consistent with the data.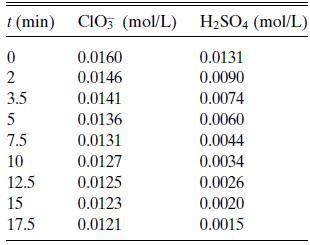
Transcribed Image Text:
t (min) CIO3 (mol/L) H₂SO4 (mol/L) 0 0.0160 0.0131 2 0.0146 0.0090 0.0141 0.0074 0.0136 0.0060 0.0044 0.0034 3.5 5 7.5 10 12.5 15 17.5 0.0131 0.0127 0.0125 0.0123 0.0121 0.0026 0.0020 0.0015
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To find a rate expression consistent with the data you can use the method of initial rates The rate ...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Propose a structure that is consistent with each set of 1H NMR data. IR data is provided for some compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) C4H1OO 6 (ppm) Splitting 1.28 1.35...
-
The formation of sugars from biomass is of considerable interest in a variety of applications, including energy alternatives to fossil fuel. Harris and Kline studied the formation of glucose from...
-
In the year to 5 April 2021, Thomas More made the following disposals: (i) A flat in a house that he had purchased on 1 December 2010 for 80,000. It had never been occupied as the main residence and...
-
For the audit of Radline Manufacturing Company, the audit partner asks you to carefully read the new mortgage contract with the First National Bank and abstract all pertinent information. List the...
-
Explain the difference between aggregate expenditure and aggregate demand.
-
Draw a frequency polygon for the frequency distribution in Example 2. Describe any patterns. Data from Example 2 Using the frequency distribution constructed in Example 1, find the midpoint, relative...
-
Congress enacted the Health Insurance Portability and Accountability Act (HIPAA) to potentially help millions of employees gain access to group health insurance. The key provision of HIPAA requires...
-
The centralized legal staff of an automotive manufacturer has determined that there is a need for an E-commerce initiative to handle the invoicing transactions received by the legal staff and the law...
-
Show that Equation 6.11a for the reaction A + B M with equal initial concentrations of A and B ( = 0) follows from Equation 6.11b for unequal initial concentrations in the limit 0. Equation 6.11a...
-
The data in Table 6.P3 are for reactant A in the irreversible reaction A + B products. The initial concentrations are believed to be in stoichiometric balance. Estimate the reaction order and find...
-
As the partner in charge of the yearly audit for CWS, you are reviewing the audit work papers for the cash and investment accounts. Before you sign off on the audit, you need to determine that...
-
3. Revised Depreciation Year #1 - Machine Cost 50,000 Salvage value 5,000 Useful Life 5 yr - use straight line depreciation - - in the beginning of year 4 add a module that increases usefulness and...
-
You are now planning to establish ABC Company which requires total investment of $8 million. Assume interest rate on borrowing (before tax) is 3%, cost of equity is 8% and tax rate is 30%. ('1)...
-
What is the price of a stock that just paid a $5 dividend yesterday and it dividends are expected to grow at a rate of 3% per annum forever? Assume that the cost of capital is 10% 2)Because of your...
-
Ricardo and Kathy Mendez divorced in 2 0 1 3 . Under the divorce decree, Ricardo is required to pay alimony of $ 3 , 0 0 0 per month and child support for their daughter, Sheri, of $ 2 , 0 0 0 per...
-
Adam would like to purchase a home. To help finance the purchase, he will borrow $300,000 from a bank. The bank will charge 6% annual interest, compounded monthly (0.005 per month). The mortgage loan...
-
In the case study of Melissa Robinson, she was able to steal over $60,000 from her employer. Why was she able to commit her fraud without detection?
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
To increase heat dissipation from a long tube with a 3.0-cm outer diameter, 12 longitudinal rectangular fins made of aluminum (k = 250 W/m K) are attached to the outer surface of the tube projecting...
-
Heat is to be provided to a small room with the finned heater sketched in Fig. P.4.1. Circulating hot oil keeps the base of the fins at 140F. The fins are made of iron with k = 40 BTU/h ft F, and the...
-
Consider a slab-shaped graphite electrode that is 3 ft long, 3 in. wide, and 0.5 in. thick. A voltage difference of 24 V is applied across the 3- ft length. Assuming that the temperature is uniform...
-
For years, Worley believed that the 7% markup covered its selling and administrative expenses and provided a reasonable profit. However, in the face of declining profits, Worley decided to implement...
-
what ways do team-based learning approaches facilitate knowledge transfer, skill development, and collective intelligence within teams, and how can organizations support continuous learning and...
-
Alex Corporation reports the following components of stockholders' equity at December 31 of the prior year. Common stock-$25 par value, 50,000 shares authorized, 30,000 shares issued and outstanding...

Study smarter with the SolutionInn App


