Calculate K for iron(II) sulfide given the following data: FeS(s) + 2e- Fe+ (aq) + 2e- Fe(s)
Question:
Calculate K for iron(II) sulfide given the following data:
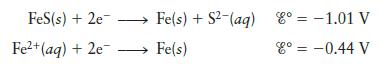
Transcribed Image Text:
FeS(s) + 2e- Fe²+ (aq) + 2e- Fe(s) + S2-(aq) = -1.01 V Fe(s) 8° = -0.44 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
reduction halfreaction FeSs2eFesS2aqEcell101V oxidation halfreac...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given the following data about the economy: a. Calculate aggregate output (GDP) and aggregate income. b. Compare the two calculations in a. Why are they not precisely equal? c. Calculate GNP. d....
-
Calculate Ksp for iron(II) sulfide given the following data: Es. cell
-
Given the following data set. Calculate the appropriate capability statistics for the process that was taken from one machine operated by one operator in the last 30 mins of his shift. Data 6.9 4.6...
-
Write a formula for a function g whose graph is similar to f(x) but satisfies the given conditions. Do not simplify the formula. f(x) = 3x 3x + 2 (a) Shifted right 2000 units and upward 70 units (b)...
-
a. In the nineteenth century, economist Alfred Marshall wrote about decreasing cost industries, writing in his Principles of Economics (available free online) that When an industry has thus chosen a...
-
Ignoring taxes in Problem 6, what is the price per share of equity under Plan I? Plan II? What principle is illustrated by your answers? Data from Problem 6 Coldstream Corp. is comparing two...
-
What happens if you specify an invalid format string?
-
Refer to the Hungry Dawg Restaurant example presented in this chapter. Health claim costs actually tend to be seasonal, with higher levels of claims occurring during the summer months (when kids are...
-
Solving via linear equation No unread replies.No replies. A small country exports soybeans and flowers. Soybeans require 8 workers per acre, flowers require 12 workers per acre, and 100,000 workers...
-
A. Russell (birthdate February 2, 1967) and Linda (birthdate August 30, 1972) Long have brought you the following information regarding their income and expenses for the current year. Russell owns...
-
The solubility product for CuI(s) is 1.1 10 -12 . Calculate the value of %8 for the half-reaction
-
Estimate for the half-reaction. 2H 2 O + 2e - H 2 + 2OH - given the following values ofG f : H 2 O(l) = -237 kJ/mol H 2 (g) = 0.0 OH - (aq) = -157 kJ/mol e - = 0.0
-
For the following telescoping series, find a formula for the nth term of the sequence of partial sums {S n }. Then evaluate to obtain the value of the series or state that the series diverges. lim S,...
-
Financial reporting in the not-for-profit sector There are no required IFRS readings for this module. Required readings from the textbook are listed with each topic. Overview This module outlines the...
-
Thanks Timothy for your input and discussing the concept of the risk-return trade-off. Under what specific circumstances can risk magnify a company's earnings in a positive way? Is it also possible...
-
1.10 Identify and discuss the circumstances where issues related to contract execution are outside of your scope of authority as an auctioneer.
-
Briefly explain what is meant by the following terms used in structuring financial products?
-
Description Amount Explanation Sales 200,000 Cost of goods sold 40,000 25,000 fixed + 18,000 variable Operating expenses 33,000 19,000 fixed + 13,000 variable Compute operating income using the...
-
How is government debt related to the government deficit? What factor contribute to a high growth rate of the debt-GDP ratio?
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
Three elements have the electron configurations 1s22s22p63s23p6, 1s22s22p63s2, and 1s22s22p63s23p64s1. The first ionization energies of these elements (not in the same order) are 0.419, 0.735, and...
-
Ionization energy is the energy required to remove an electron from an atom in the gas phase. The ionization energy of gold is 890.1 kJ/ mol. Is light with a wavelength of 225 nm capable of ionizing...
-
Predict some of the properties of element 117 (symbol Uus following conventions proposed by the International Union of Pure and Applied Chemistry [IUPAC]). a. What will be its electron configuration?...
-
Assume that you will borrow $ 4 0 0 , 0 0 0 from a bank that charges 4 . 5 percent interest compounded monthly and repay the loan with monthly equal payments at the end of each month over the next 1...
-
How do endocrine glands integrate diverse physiological signals and regulate systemic homeostasis through the secretion of hormones, coordinating intricate intercellular communication and adaptive...
-
How do circadian rhythms, governed by the central circadian clock and peripheral oscillators within endocrine tissues, regulate the temporal patterns of hormone secretion, tissue responsiveness, and...

Study smarter with the SolutionInn App


