Consider the nitration of the compound If the reaction can be controlled so that one NO 2
Question:
Consider the nitration of the compound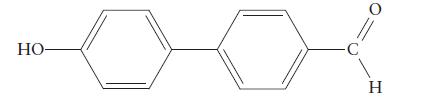 If the reaction can be controlled so that one NO2 group replaces one H atom of the molecule, where do you expect the nitro group to end up in the product?
If the reaction can be controlled so that one NO2 group replaces one H atom of the molecule, where do you expect the nitro group to end up in the product?
Transcribed Image Text:
H 0 HO-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The image shows a compound with two benzene rings connected by a single bond with one ring having a ...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The formal study of probability began with questions regarding gambling and games of chance. The conventional analysis of gambling is based on the expected values of these games which is always...
-
The presence of additional nitro groups can have an impact on the temperature at which a nucleophilic aromatic substitution will readily occur. Consider the following example. When both R groups are...
-
Consider Figs. 16.3 and 16.4 illustrating cascade control. (a) Suppose you were to apply feedforward control, instead of cascade control, to handle disturbances D 1 and D 2 . Where do you expect...
-
Suppose the comparative balance sheets of Sage Hill Inc.. are presented here. SAGE HILL INC. Condensed Balance Sheet May 31 ($ in millions) 2019 2018 Assets Current Assets $9,680 $8,760 Property,...
-
Using loop analysis and MATLAB, find Io in the network infigure. 12/0v 2/0 A j1a 4/0 A 320 -j2 0
-
Suppose that an investor holds a share of Sophia common stock, currently valued at $50. She is concerned that over the next few months the value of her holding might decline, and she would like to...
-
This exercise considers the data described in the Section 13.2.2 ratemaking classification example using data in Table 13.3 Table 13.3 . a. Fit a gamma regression model using a log-link function with...
-
Sean McNamee was the owner of an accounting firm, W. F. McNamee & Co., LLC (WFM), which he founded and formed in Connecticut as a limited liability company (LLC). For federal tax purposes, an LLC can...
-
Problem #2\ Erica, Deana & Tara had average capital balances of $350,000,$380,000 and $400,000 respectively during the current fiscal year. The partnership agreement provides for an allowance of 4%...
-
Identify the type and number of bonds on carbon atom 2 in (a) Pentane; (b) 2-pentene; (c) 2-pentyne.
-
Give the systematic name of (a) CH 3 COOH; (b) CH 3 CH 2 CH 2 COOH; (c) CH 2 (NH 2 )COOH.
-
Roots to Branches Corporation, a company specializing in event dcor, is considering two mutually exclusive investments: Roots and Branches. Both investments will assist the company with the dcor...
-
How would you define the most "hardened of criminals" or the "worst of the worst"?
-
What is the reason for the absence of a land area requirement for barangays? and What are the two positions that are not mandatory for cities and municipalities? explain.
-
How do you hold yourself and your peers accountable to the GCU Code of Conduct and Academic Integrity standards?
-
Management of Anthony Walker, a confectioner, is considering purchasing a new jelly bean-making machine at a cost of $282,730. They project that the cash flows from this investment will be $103,710...
-
Information about an entity's financial position is primarily provided in: 1 . The income statement 2 . The statement of financial position 3 . Retained earnings 4 . The statement of cash flows
-
Abby is the sole owner of a nail salon. Her costs for a manicure are given by TC = 10 + q2 AC = 10/q + q MC = 2q The nail salon is open only 2 days a weekWednesdays and Saturdays. On both days, Abby...
-
Identify the tax issues or problems suggested by the following situations. State each issue as a question. Jennifer did not file a tax return for 2007 because she honestly believed that no tax was...
-
Draw a bondline structure showing the zwitterionic form of each of the following amino acids: (a) l-Valine (b) l-Tryptophan (c) l-Glutamine (d) l-Proline
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
Draw a Fischer projection for each of the following amino acids: (a) l-Threonine (b) l-Serine (c) l-Phenylalanine (d) l-Asparagine
-
1. Consider the two-period binomial model with a non-dividend paying stock S, where So = 4, u = 2, d = 0.5, r = 0.25. Your boss has asked you to price an MaxCall (Lookback) option which expires at...
-
1. Consider the following one-period binomial model. Assume the initial value of the stock is So. The stock price will go to either Sou or Sod at time t = 1. Assume that the one-period simple...
-
(a) The following table shows the timing constraints for the three tasks, i.e. T1, T2, and T3. Perform the scheduling (from time t= 0 until 20) for the three tasks using the Earliest Deadline First...

Study smarter with the SolutionInn App


