Estimate the vapor pressure of heavy water, D 2 O, and of normal water at 25C by
Question:
Estimate the vapor pressure of heavy water, D2O, and of normal water at 25°C by using data in Appendix 2A. How do these values compare with each other? Using your knowledge of intermolecular forces and any quantum effects, such as the zero point energy of intermolecular vibrations, explain the reason for the difference observed.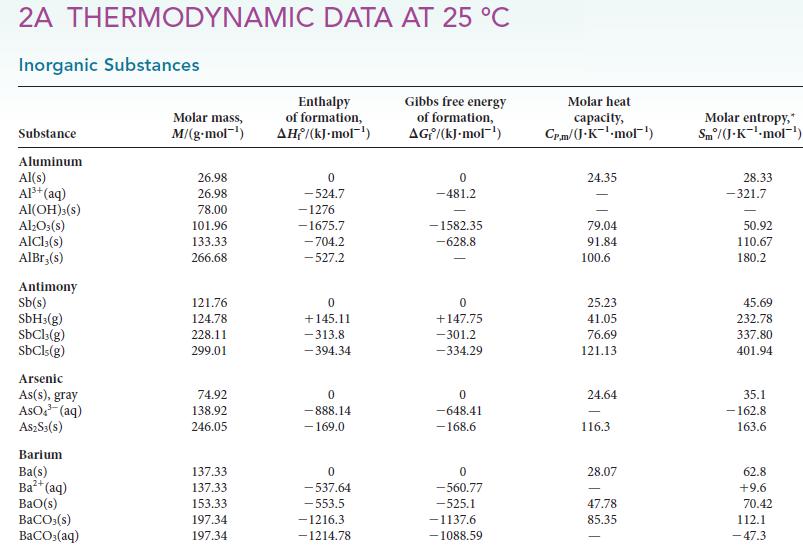
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g-mol ¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/(J.K-¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy, Sm/(J.K¹-mol-¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
For HO PH0 0...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Appendix B lists the vapor pressure of water at various external pressures. (a) Plot the data in Appendix B, vapor pressure (torr) versus temperature (oC). From your plot, estimate the vapor pressure...
-
In Section 11.5 we defined the vapor pressure of a liquid in terms of an equilibrium. (a) Write the equation representing the equilibrium between liquid water and water vapor and the corresponding...
-
Calculate the vapor pressure of water at 150C using the van der Waals equation of state. Repeat the calculation at 370C. How do these values compare with values obtained with the IF97 model?
-
The table shows equivalent temperatures in degrees Celsius and degrees Fahrenheit. (a) Plot the data with Fahrenheit temperature on the x-axis and Celsius temperature on the y-axis. What type of...
-
The diffusivity of toluene in air was determined experimentally by allowing liquid toluene to vaporize isothermally into air from a partially filled vertical tube 3 mm in diameter. At a temperature...
-
a. Use the random numbers 0.3753, 0.9218, 0.0336, 0.5145, and 0.7000 to generate five simulated values for the PortaCom direct labor cost per unit. b. Use the random numbers 0.6221, 0.3418, 0.1402,...
-
Use Table 1, or software, to find (a) \(B(7 ; 18,0.45)\); (b) \(b(7 ; 18,0.45)\); (c) \(B(8 ; 11,0.95)\); (d) \(b(8 ; 11,0.95)\); (e) \(\sum_{k=4}^{11} b(k ; 11,0.35)\); (f) \(\sum_{k=2}^{4} b(k ;...
-
Mrs. O is negotiating to purchase a tract of land from DC Company, which is a calendar year taxpayer. DC bought this land six years ago for $480,000. According to a recent appraisal, the land is...
-
1. A circuit has three resistors connected to a 3.00-V battery in series: R = 15.02, R2 10.02, and R3 = 25.02. Find the equivalent resistance, Req, the total current in the circuit, Itot, and the...
-
You recently took 5,000 square feet of new space that hadn't been built out at all and put in a childcare facility. That project ended up costing $550,000. But, for this project, you have to demo the...
-
A reaction vessel is filled with Cl 2 (g) at 1.00 bar and Br 2 (g) at 1.00 bar, which are allowed to react at 1000. K to form BrCl(g)according to the equation Br 2 (g) + Cl 2 (g) 2 BrCl(g), K = 0.2....
-
The gas phosphine, PH 3 , decomposes by the reaction 2 PH 3 (g) 2 P(s) + 3 H 2 (g). In an experiment, pure phosphine was placed in a rigid, sealed flask of volume 1.00 L at 0.64 bar and 298 K. After...
-
An executive from a large merchandising firm has called your vice president for production to get a price quote for an additional 100 units of a given product. The vice president has asked you to...
-
Maintenance on a test track simulator used to exercise vehicles 24/7 for engineering reliability analyses is expected to require $14,000 the first year increasing by 10 percent each year thereafter...
-
You decide to open a retirement account at your local bank that pays 8 percent/year/month (8 percent per year compounded monthly). For the next 20 years, you will deposit $400 per month into the...
-
Using real-word examples, discuss the extent to which demand-side policies are effective in reducing inflation.
-
One of the arguments for punitive damages (damages paid to the plaintiff in excess of actual damages) is that such damage payments compensate for the fact that lawsuits are not always filed, even if...
-
Does the problem of monopoly provision of a bad arise with a true Pigovian fee? Why?
-
ABC Company purchases all the assets of Johns Saw Shop. Details on basis and fair market values of Johns Saw Shops assets are as follows: a. What is ABCs basis in the assets purchased if ABC pays...
-
In Exercises 516, find the focus and directrix of the parabola with the given equation. Then graph the parabola. y 2 = 4x
-
Propose the structure of a compound that exhibits the following 1 H NMR data: (a) C 5 H 10 O 1.09 (6H, doublet) 2.12 (3H, singlet) 2.58 (1H, septet) (b) C 5 H 12 O 0.91 (3H, triplet) 1.19 (6H,...
-
A compound with molecular formula C 8 H 10 O produces six signals in its 13 C NMR spectrum and exhibits the following 1 H NMR spectrum. Deduce the structure of the compound. Proton NMR Chemical Shift...
-
Deduce the structure of a compound with molecular formula C 9 H 12 that produces the following 1 H NMR spectrum: Proton NMR Chemical Shift (ppm)
-
HTBM Ltd. has an outstanding issue of bond with a par value of $1,000, paying 8 percent coupon rate semi-annually. And, the company just paid a dividend of $2.70 per share. The dividends are expected...
-
Over the past several years, Mr Lee has been able to save regularly. As a result, today he has $14,188 savings. He wants to establish her retirement fund in 5 years and feels he will need $50,000 to...
-
A 30-year fully amortizing mortgage loan was made 10 years ago for $75,000 at 10 percent interest. The borrower would like to prepay the mortgage balance by $10,000. Assuming he can reduce his...

Study smarter with the SolutionInn App


