A compound with molecular formula C 8 H 10 O produces six signals in its 13 C
Question:
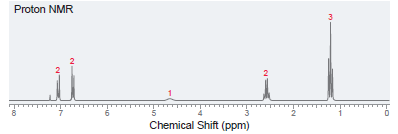
Transcribed Image Text:
Proton NMR Chemical Shift (ppm) -00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the compound with molecular formula C8H10O that gives the IR and 1H NMR spectra shown in Figure 14.23. 23 16 27 2 29 35 13 14 15 16 800 200 2400 200 0 6 (ppm) frequency
-
A compound with molecular formula C 8 H 10 produces three signals in its 13 C NMR spectrum and only two signals in its 1 H NMR spectrum. Deduce the structure of the compound.
-
A compound with molecular formula C 8 H 10 produces an IR spectrum with many signals, including 3108, 3066, 3050, 3018, and 1608 cm -1 . The 1 H NMR spectrum of this compound exhibits a singlet at...
-
Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20
-
What are three drawbacks of mobile and wireless networks?
-
Consider a potential that depends only on the radial direction \(r\) in a three-dimensional space, \(V(r)\), with \(V(r\) such that \( ightarrow \infty)=0\) while \(V(0)=-V_{0} <0\) and \(V_{\max...
-
Table B. 24 contains data on home rental prices and home sales. Use all possible regressions to build a model for these data. Perform a residual analysis on the final model and comment on model...
-
The consolidated statements of cash flows and related note disclosure for Le Chteau Inc. are in Exhibits 5.23A and 5.23B. EXHIBIT 5.23A Le Chteau Inc.s 2017 Consolidated Statements of Cash Flows...
-
2.) What is the specific goal of this lab? Exactly what question are you trying to answer? Be as specific as possible. ("To learn about topic X..." is not specific!) 3.) What specific measurements or...
-
Let Ai be the set of all nonempty bit strings (that is, bit strings of length at least one) of length not exceeding i. Find (a) (b) UA
-
Propose the structure of a compound that exhibits the following 1 H NMR data: (a) C 5 H 10 O 1.09 (6H, doublet) 2.12 (3H, singlet) 2.58 (1H, septet) (b) C 5 H 12 O 0.91 (3H, triplet) 1.19 (6H,...
-
What type of symbiotic relationshipmutualism, commensalism, or predationdo you think exists between the pygmy seahorse and the gorgonian coral pictured in Figure 5.22? Explain your answer.
-
One of the CRASH variables saved in the accompanying file is the severity of a driver's head injury when the car is in a head-on collision with a fixed barrier while traveling at 35 miles per hour....
-
A. B. A business, electricity charges amount to RM12,000 per annum. In the year to 31 December 2021, RM9,000 has been paid. The electricity for the final quarter is paid in January 2022. Show the...
-
A $1000 increase in real per capita GNP will cause the human development index (HDI) to what? Explain.
-
Can you elaborate on the role of goal setting theory and expectancy theory in shaping performance management processes, examining how clear, challenging goals and performance incentives can motivate...
-
A decedent's interest and taxes are deductible on both Forms 706 and 1041 only if: A. The decedent's will did not provide instructions. B. Either item is a deduction in respect of a decedent (DRD)....
-
Describe and provide examples of price and quality controls. Price Control is a policy that sets or caps the price of services or the growth in prices directly. It can be used to limit health care...
-
Castile Inc. had a beginning balance of $4,000 in its Accounts Receivable account. The ending balance of Accounts Receivable was $4,500. During the period, Castile recognized $68,000 of revenue on...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
Name the following alkenes, including the cis or Tran?s designation: (b)
-
Which member in each of the following sets has higher priority? (a) H or Br (b) C1 or Br (c) CH 3 or CH 2 CH 3 (d) NH 2 or OH (e) CH 2 OH or CH 3 (f) CH 2 OH or CH = O
-
Rank the following sets of substituents in order of Cahn-In-gold Prelog priorities: (a) CH3, 0H, H, C1 (b) CH3, CH2CH3, CH = CH2, CH2OH (c) CO2H, CH2OH, C = N, CH2NH2 (d) CH2CH3, C = CH, C = N,...
-
Estimate the final temperature in Celsius of the combination if I drop 5 rolls of pennies at 2 0 C ( each roll is 5 0 pennies stacked together; assume they are pure copper ) into a cup of recently -...
-
A 100 gram mass is hung of then end of a cantilever beam . The beam is made of brass, and is 30 cm long (), 2 cm wide, and 3 mm thick. Find the resulting deflection ()
-
The nitrogen-vacancy (NV) and silicon-vacancy (SiV) defects in diamond can lead to many exciting quantum nanotechnologies in physical and biological sciences, including single-photon sensors,...

Study smarter with the SolutionInn App


