Ethanol is a renewable and clean-burning component of gasoline: What is the change in internal energy for
Question:
Ethanol is a renewable and clean-burning component of gasoline: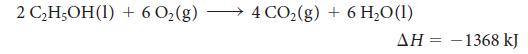
What is the change in internal energy for the reaction of 1.00 mol C2H5OH(l)?
Transcribed Image Text:
2 CHOH(I) + 602(g) - 4 CO2(g) + 6 H,O(1) ΔΗ -1368 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The change in internal energy AH for the given reaction is 1368 kJmol The relationship between the c...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
A sample consisting of 22.7 g of a nongaseous, unstable compound X is placed inside a metal cylinder with a radius of 8.00 cm, and a piston is carefully placed on the surface of the compound so that,...
-
The standard enthalpy of formation of H2O(l) at 298 K is 285.8 kJ/ mol. Calculate the change in internal energy for the following process at 298 K and 1 atm: H2O(l) H2(g) + O2(g) Eo = ?
-
Violet Flowers expects to sell 3,000 plants a month. She estimated the following monthly costs: Variable Costs $9,000 Fixed Costs $15,000 During her second month of operation, Violet would like to...
-
SEK Printing provides printing services to many different corporate clients. Although SEK bids most jobs, some jobs, particularly new ones, are negotiated on a cost-plus basis. Cost-plus means that...
-
In searching for a boat lost at sea, a Coast Guard cutter leaves a port and travels 75.0 mi due east. It then turns 65 north of east and travels another 75.0 mi, and finally turns another 65.0 toward...
-
(a) In Figure 7.5, what is the momentum of the ball during the collision? (b) Is the momentum of the ball constant before, during, and after the collision? If so, why? If not, why not, and for what...
-
1. Identify where (in its value system) and how IKEA have achieved cost leadership. 2. Identify how IKEA have achieved differentiation from their competitors. 3. Explain how IKEA tries to ensure that...
-
Question 11pts The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed...
-
Assuming that the heat capacity of an ideal gas is independent of temperature, calculate the entropy change associated with lowering the temperature of 4.10 mol of ideal gas atoms from 225.71C to...
-
The bond enthalpy in NO is 632 kJ mol 1 and that of each NO bond in NO 2 is 469 kJ mol 1 . Using Lewis structures and the data in Table 4E.3, explain (a) The difference in bond enthalpies between...
-
What products would you expect from the reaction of propyllithium (CH3CH2CH2Li) with each of the following reagents? (a) (b) (c) (d) Ethanol (e) H, then H3O , then NH,CI, H20 1-Pentyne, then , then...
-
Ron is a director of Standard Company. Ron has a right to a. compensation for his efforts on Standards behalf. b. transfer shares of Standard stock. c. participate in Standard board meetings. d....
-
Unlike managers, officers are not corporate employees. (True/False)
-
Andrei rents an apartment from Sue. Two months later, Andrei moves out and arranges with Lee for Lee to move in and pay the rent to Sue for the rest of the term. This is a. an assignment. b. a...
-
What distinguishes a fixed-term tenancy from a periodic tenancy?
-
U.S. Goods, Inc. (USG) recruits workers from other countries to work in its U.S. plant. To comply with the Immigration Act, USG must show a. that there is a shortage of qualified U.S. workers to...
-
For each of the following goods, indicate whether they are nonrival and/or nonexclusive. Indicate whether they are private or public goods. a. Hot dogs b. Cable TV c. Broadcast TV d. Automobiles e....
-
On October 1, 2021, Adoll Company acquired 2,600 shares of its $1 par value stock for $38 per share and held these shares in treasury. On March 1, 2023, Adoll resold all the treasury shares for $34...
-
A sample of S8(g) is placed in an otherwise empty, rigid container at 1325 K at an initial pressure of 1.00 atm, where it decomposes to S2(g) by the reaction S8(g) 4S2(g) At equilibrium, the partial...
-
At a particular temperature, 12.0 moles of SO3 is placed into a 3.0- L rigid container, and the SO3 dissociates by the reaction 2SO3(g) 2SO2(g) + O2(g) At equilibrium, 3.0 moles of SO2 is present....
-
At 900oC, Kp = 1.04 for the reaction CaCO3(s) CaO(s) + CO2(g) At a low temperature dry ice (solid CO2), calcium oxide, and calcium carbonate are introduced into a 50.0- L reaction chamber. The...
-
. 1 2 3 4 5 5 7 B 9 1-Oct Santana Rey invested $45,000 cash, a $20,000 computer system, and a $8,000 of office equipment in the company. Go to Accounting, Transactions, More and Add Journal...
-
After the success of the company's first two months, Santana Rey continues to operate Business Solutions. The November 30, 2021, unadjusted trial balance of Business Solutions (reflecting its...
-
Preparing a consolidated income statement - with noncontrolling interest, but no AAP or intercompany profits A parent company purchased an 80% interest in its subsidiary several years ago with no AAP...

Study smarter with the SolutionInn App


