Identify the hybridization of each carbon and nitrogen atom in caffeine (see Exercise 11.25c). Exercise 11.25c Identify
Question:
Identify the hybridization of each carbon and nitrogen atom in caffeine (see Exercise 11.25c).
Exercise 11.25c
Identify all the functional groups in each of the following compounds:
(c) caffeine, the stimulant in coffee, tea, and cola drinks,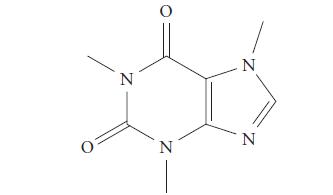
Transcribed Image Text:
Z Z - Z Z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Hybridization in molecules such as caffeine is often determined by looking at the number of bonds sigma bonds and lone pairs around a particular atom ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify the hybridization of each carbon and nitrogen atom in guanine (see Exercise 11.23a). Exercise 11.23a Write the chemical formula of the compound represented by each of the following line...
-
Two compounds with the formula CH 3 -CH=N-CH 3 are known. (a) Draw a Lewis structure for this molecule, and label the hybridization of each carbon and nitrogen atom. (b) What two compounds have this...
-
a. What is the hybridization of each of the carbon atoms in the following compound? b. What is the hybridization of each of the carbon, oxygen, and nitrogen atoms in the following compounds? CH CHCH...
-
Being able to identify an industry to use for benchmarking yourfirm's results with similar companies is frequently not easy. Choose a type of business and go to www.naics.com. This Web site allows...
-
Given the series circuit in figure, find the value of R so that the BW of the network about the resonant frequency is 200rad/s. (a) 8 ? (b) 2 ? (c) 4 ? (d) 6 ? 20 mH ww vs(1) 50 F
-
The stock market crash of 1929 and the Great Depression that followed were caused in part because so many investors blindly put their money into stocks they knew nothing about. During the 1920s, it...
-
Assuming a 10 percent annual discount rate, which system should EAMO purchase? Cost Estimates for EOP and PMS Year EOP ($) PMS ($) 0 (the current year) 1,500,000 750,000 1 100,000 300,000 2 100,000...
-
Swain Enterprises issues a 60-day note for $800,000 to Hill Industries for merchandise inventory. Hill Industries discounts the note at 6%. a. Journalize Swain Enterprises' entries to record: 1. The...
-
Caspian Sea Drinks is considering the purchase of a plum juicer - the PJX 5 . There is no planned increase in production. The PJX 5 will reduce costs by squeezing more juice from each plum and doing...
-
Draw the structure of the principal product formed from each pair of reagents in condensation reactions: (a) Butanoic acid with 2-propanol; (b) Ethanoic acid with 1-pentanol; (c) Hexanoic acid with...
-
Indicate which of the following molecules are optical isomers and identify the chiral carbon atoms in those that are: (a) CH 3 CHBrCH 2 Br; (b) CH 3 CH 2 CHClCH 2 CH 3 ; (c) 2-bromo- 2-methylpropane;...
-
1. What could be the main motives for Starbucks in owning most of its coffee houses compared with other entry modes and operation forms? 2. How does Starbucks entry into the grocery market affect the...
-
Below are incomplete financial statements for Hurricane, Incorporated Required: Calculate the missing amounts. Complete this question by entering your answers in the tabs below. Income Statement Stmt...
-
TBTF Incorporated purchased equipment on May 1, 2021. The company depreciates its equipment using the double-declining balance method. Other information pertaining to the equipment purchased by TBTF...
-
Coco Ltd. manufactures milk and dark chocolate blocks. Below is the information relating to each type of chocolate. Milk Chocolate Selling price per unit $6 Variable cost per unit $3 Sales mix 4 Dark...
-
Data related to 2018 operations for Constaga Products, a manufacturer of sewing machines: Sales volume 5,000 units Sales price $300.00 per unit Variable production costs Direct materials 75.00 per...
-
6. (20 points) Sections 3.1-3.5, 3.7 Differentiate the following functions, state the regions where the functions are analytic. a. cos(e*) b. 1 ez +1 c. Log (z+1) (Hint: To find where it is analytic,...
-
If a solution containing 0.10 M Cl-, Br-, I-, and CrO42- is treated with Ag+, in what order will the anions precipitate?
-
Data 9.2 on page 540 introduces the dataset Cereal, which includes information on the number of grams of fiber in a serving for 30 different breakfast cereals. The cereals come from three different...
-
A central issue in the design of aircraft is improving the lift of aircraft wings. To assist in the design of more efficient wings, wind-tunnel tests are performed in which the pressures at various...
-
Oxygen sensing is important in biological studies of many systems. The variation in oxygen content of sapwood trees was measured by del Hierro and coworkers [ J. Experimental Biology 53 (2002): 559]...
-
The pyrene/coumarin FRET pair (r 0 = 39 ) is used to study the fluctuations in enzyme structure during the course of a reaction. Computational studies suggest that the pair will be separated by 35 ...
-
Choose two stocks from the same industry to minimize the influence of other confounding factors. You choose the industry that you are relatively more familiar with, and then estimate the implied...
-
why should Undertake research to review reasons for previous profit or loss?
-
A pension fund's liabilities has a PV01 of $200 million. The plan has $100 billion of assets with a weighted average modified duration of 8. The highest duration bond that the plan can invest in has...

Study smarter with the SolutionInn App


