Indium arsenide crystallizes in the zinc-blende ( sphalerite) structure (Fig. 3H.32). (a) What are the coordination numbers
Question:
Indium arsenide crystallizes in the zinc-blende ( sphalerite) structure (Fig. 3H.32).
(a) What are the coordination numbers of the indium and arsenide ions?
(b) What is the formula of indium arsenide?
FIGURE 3H.32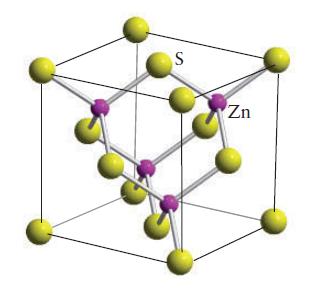
Transcribed Image Text:
S Zn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a Indium arseni...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Silver iodide crystallizes in the zinc blende structure. The separation between nearest-neighbor cations and anions is approximately 325 pm, and the melting point is 558 C. Cesium chloride, by...
-
Copper iodide crystallizes in the zinc blende structure. The separation between nearest-neighbor cations and anions is approximately 311 pm, and the melting point is 606 C. Potassium chloride, by...
-
Which compound is most likely to crystallize in the zinc blende structure? (a) RbCl (Rb + radius = 148 pm; Cl radius = 181 pm) (b) MgCl 2 (Mg 2+ radius = 65 pm; Cl radius = 181 pm) (c) CuI (Cu +...
-
For the following exercises, use shells to find the volume generated by rotating the regions between the given curve and y = 0 around the x-axis. 130. y = 1-x,x = 0, and x = 1 131. y = x, x = 0, and...
-
Two couples act on the frame. If d = 4 ft, determine the resultant couple moment. Compute the result by resolving each force into x and y components and (a) Finding the moment of each couple (Eq....
-
Determine the domain and the range of the function shown in the figure at right. LLUT + YA 5432 --- 3.. 1 -5-4-3-2-1 1 2 3 4 5 VA - -4 -5 X
-
You are working on charge-storage devices for a research center. Your goal is to store as much charge on a given device as possible. The facilities allow you to generate almost any potential...
-
Tomiko owns the copyright to several classic Motown songs. In 2009, he became aware that Tinseltown Records was selling several of his songs without his permission. He sues Tinseltown seeking...
-
6. An annular disk of mass M, inner radius a and outer radius b is placed on a horizontal surface with coefficient of friction , as shown in the figure. At some time, an impulse Jox is applied at a...
-
Air bags in automobiles contain sodium azide, NaN 3 , which decomposes rapidly during a collision to give nitrogen gas and sodium metal. The nitrogen gas liberated by this process instantly inflates...
-
The effect of high pressure on organisms, including humans, is studied to gain information about deep-sea diving and anesthesia. A sample of air occupied 1.00 L at 25C and 1.00 atm. What pressure (in...
-
Scottsdale Ltd. manufactures a variety of high-volume and low-volume products to customer demand. Presented is information on 2009 manufacturing overhead and activity cost drivers. Product XI...
-
Generally speaking, what happens to the value of a countrys currency when imports exceed exports and how, in terms of supply and demand, does this take place?
-
Explain how having a single type of aircraft in a fleet affects an airlines ability to recover from irregular operations, such as diversion recovery or an extended airport shutdown due to bad weather?
-
Explain why airlines originally formed alliances with foreign carriers.
-
What do you conclude about the gains from trade when you compare the sum of producer surplus and government gain to the loss of consumer surplus?
-
From time to time, when purchasing certain items such as jewelry, the salesperson will ask the customer what price the customer has in mind. What do you suppose is the purpose of this question?
-
List and describe the three control strategies proposed for IDPS control.
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
Calculate Ksp for iron(II) sulfide given the following data: Es. cell
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Au3+] = 1.0 Ã 102 M and [Tl+] = 1.0...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Au3+] = 1.0 Ã 102 M and [Tl+] = 1.0...
-
Identify the relevant costs associated with each of Nancy's three options. Total average monthly cost for new used and leased.
-
Use the information below to prepare the operating section of a cash flow statement using the indirect method. (24 points) DEF, Inc. Comparative Balance Sheets As of December 31, 2022 and 2021 2022...
-
Create a balance sheet Total Project Budget: $745,700 Technology Development $ Virtual Platform Development 50,000.00 Software integration and customization 20,000.00 Server hosting and maintenance...

Study smarter with the SolutionInn App


