Methanol (CH 3 OH) is sometimes used as a fuel in high-performance engines. Using the data in
Question:
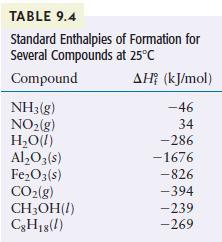
Methanol (CH3OH) is sometimes used as a fuel in high-performance engines. Using the data in Table 9.4, compare the standard enthalpy of combustion per gram of methanol with that of gasoline. Gasoline is actually a mixture of compounds, but assume for this problem that gasoline is pure liquid octane (C8H18).
Transcribed Image Text:
TABLE 9.4 Standard Enthalpies Several Compounds at 25C Compound NH3(g) NO(g) HO(1) AlO3(s) FeO3(s) CO(g) CHOH(1) C8H18(1) of Formation for AH (kJ/mol) -46 34 -286 -1676 -826 -394 -239 -269
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The combustion reaction for methanol is 2CHOH1 30g 2COg 4HOl Using the standard enthalpies of format...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Methanol (CH3OH) has also been proposed as an alter-native fuel. Calculate the standard enthalpy of combustion per gram of liquid methanol, and compare this answer to that for ethanol in Exercise 80.
-
Methanol (CH3OH) is used as a fuel in race cars. (a) Write a balanced equation for the combustion of liquid methanol in air. (b) Calculate the standard enthalpy change for the reaction, assuming...
-
In January 1995, the Office of University Evaluation at Arizona State University surveyed faculty and staff members to find out their reaction to the closure of the university during Winter Break,...
-
Glass bottles can be either recycled (crushed and re-melted) or reused. The market will tend to choose the cheapest path. What factors will tend to affect the relative cost of these options? Is the...
-
Let the value of a in the logistic equation, Equation 4.46, be equal to 0.9, Make a map like that in Figure 4-21 when x1 = 0.4. Make the plot for three other values of x1 for which 0 < x1 < 1.
-
Suppose that tables T1 and T2 have a 1:N relationship. Show the SQL statements necessary to fill an intersection T1_T2_INT. Make up your own names for primary and foreign keys.
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
McLennon Company has a plant capacity of 100,000 units per year, but its budget for this year indicates that only 60,000 units will be produced and sold. The entire budget for this year is as...
-
Mercedes-Benz AG is a subsidiary of Daimler AG known for producing luxury vehicles and commercial vehicles. This company has strongly pushed for product placement in Hollywood movies. In 2015,...
-
Assuming that the combustion of hydrogen gas provides three times as much energy per gram as gasoline, calculate the volume of liquid H 2 (density = 0.0710 g/mL) required to furnish the energy...
-
Using the standard enthalpies of formation listed in Table 9.4, calculate the standard enthalpy change for the overall reaction that occurs when ammonia is burned in air to form nitrogen dioxide and...
-
Professional auditing standards identify the principal management assertions that underlie a set of financial statements. What were the key assertions that Arthur Young should have attempted to...
-
a municipal securities dealer may consider all of the following in determining a fair and reasonable price for a municipal security, except: A. The best judgment of the dealer as to the value of the...
-
The marketing and underwriting teams at Shelby Insurance have decided that they need some new forms and endorsements to address the growing cyber risk loss exposure. Which one of the following...
-
Inflation refers to the sustained boom inside the standard charge stage of merchandise and services through the years. As a financial corporation, inflation influences our clients' funding...
-
1.Based on your knowledge concerning capital budgeting, discuss several factors that would increase the risk associated with a company's capital investment decision. In your opinion explain which...
-
Accountants use the following to help ensure they use standardized approaches when making accounting decisions (example: how to account for a specific cost item) , Question 7 options: decision making...
-
Rex Santos, a cost accountant, prepares a product profitability report for Jane Gates, the production manager. Much to Rexs surprise, almost one-third of the companys products are not profitable. He...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
Write chemical equations for (a) The burning of lithium in oxygen; (b) The reaction of sodium metal with water; (c) The reaction of fluorine gas with water; (d) The oxidation of water at the anode of...
-
Identify the oxidation number of the halogen in (a) Hypoiodous acid; (b) ClO 2 ; (c) Dichlorine heptoxide; (d) NaIO 3 .
-
What are the sources for the production of helium and argon?
-
Arcelor Mittal Dofasco Inc. is a highly successful international steel company. They work together with the communities they operate in to decide what projects will improve the local environment and...
-
In a university stationery store, pencils are sold at a constant rate of 6 0 per week ( the stationery store works 5 2 weeks a year ) . The stationery store costs $ 0 . 0 2 each and sells them for $...
-
1. To what degree did that happen as she rose within the corporate structure? Also, what changed? 2. According to Sandberg, how and why are women in leadership judged through a different lens than...

Study smarter with the SolutionInn App


