Predict the standard potential of each of the following galvanic cells: 3+ (a) Pt(s)| Fe+ (aq), Fe+
Question:
Predict the standard potential of each of the following galvanic cells: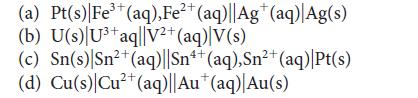
Transcribed Image Text:
3+ (a) Pt(s)| Fe³+ (aq), Fe²+ (aq)||Ag* (aq) Ag(s) (b) U(s) U³+ aq||V²+ (aq) V(s) 2+ (c) Sn(s) Sn²+ (aq)||Sn4+ (aq),Sn²+ (aq)|Pt(s) 2+ (d) Cu(s) Cu²+ (aq)||Au* (aq)| Au(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
cell To predict the standard cell potential El for each galvanic cell we can use the standard reduct...View the full answer

Answered By

Mwangi Clement
I am a tried and tested custom essay writer with over five years of excellent essay writing. In my years as a custom essay writer, I have completed more than 2,000 custom essays in a diverse set of subjects. When you order essays from me, you are working with one of the best paper writers on the web. One of the most common questions I get from customers is: “can you write my essay?” Upon hearing that request, my goal is to provide the best essays and overall essay help available on the web. I have worked on papers in subjects such as Nursing and Healthcare, English Literature, Sociology, Philosophy, Psychology, Education, Religious Studies, Business, Biological Sciences, Communications and Media, Physical Sciences, Marketing and many others. In these fields, my specialties lie in crafting professional standard custom writings. These include, but are not limited to: research papers, coursework, assignments, term papers, capstone papers, reviews, summaries, critiques, proofreading and editing, and any other college essays.
My extensive custom writings experience has equipped me with a set of skills, research abilities and a broad knowledge base that allows me to navigate diverse paper requirements while keeping my promise of quality. Furthermore, I have also garnered excellent mastery of paper formatting, grammar, and other relevant elements. When a customer asks me to write their essay, I will do my best to provide the best essay writing service possible. I have satisfactorily offered my essay writing services for High School, Diploma, Bachelors, Masters and Ph.D. clients.
I believe quality, affordability, flexibility, and punctuality are the principal reasons as to why I have risen among the best writers on this platform. I deliver 100% original papers that pass all plagiarism check tests (Turnitin, Copyscape, etc.). My rates for all papers are relatively affordable to ensure my clients get quality essay writing services at reasonable prices.
4.50+
5+ Reviews
14+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the standard potential of each of the following galvanic cells: 3+ (a) Pt(s) Cr+ (aq), Cr+ (aq)||Cu+ (aq) |Cu(s) (b) Ag(s) AgI(s) I (aq)||CI (aq)|AgCl(s) Ag(s) (c) Hg(1) HgCl (s) CIT(aq) |Hg+...
-
A galvanic cell has the following cell reaction: M(s) + 2 Zn 2+ (aq) 2 Zn(s) + M 4+ (aq). The standard potential of the cell is 10.16 V. What is the standard potential of the M 4+ /M redox couple?
-
Sketch the galvanic cells based on the following half-reactions. Calculate Ïo, show the direction of electron flow and the direction of ion migration through the salt bridge, identify the...
-
Built-Tite uses job order costing. The T-account below summarizes Factory overhead activity for the current year. Factory Overhead Debit Credit 16,200 106,600 25,200 60,200 1. Compute total applied...
-
Weighted-average method, inspection at 80% completion (chapter appendix). (A. Atkinson) The Kim Company is a furniture manufacturer with two departments: molding and finishing. The company uses the...
-
Keith Whittingham, as manager of Designs by Whittingham, is upgrading his CAD software. The high-performance (HP) software rents for $3,000 per month per workstation. The standard-performance (SP)...
-
Differentiate between Type I and Type II errors.
-
Primus Corp. is planning to convert an existing warehouse into a new plant that will increase its production capacity by 45 percent. The cost of this project will be $7,125,000. It will result in...
-
Mark opens his own company and completes the following transactions in May: 5/1 Mark invests $14,000 into the business. 5/3 Purchased $3,800 of equipment on account. 5/4 Paid $560 premium for a...
-
A careless laboratory technician prepares 300.0 mL of 0.0175 m KOH(aq) and pipets 25.0 mL of the solution into a beaker. The beaker is allowed to stand in a warm place for two days before use, during...
-
The molar solubility of silver sulfite, Ag 2 SO 3 , is 1.55 * 10 5 mol L 1 . What is the K sp of silver sulfite?
-
What is the relative importance of labor as an economic resource?
-
The Assignment A for this Workshop is to perform an analysis which, given a Treasury yield curve, computes the corresponding spot rates. You will be working with daily yield data for 2021. In all...
-
The aims of this experiment are as follow: Calculate the volume % of each of the following component based on the information in the below tables. Calculate the weight percentage of the each of the...
-
. In Eq. (4-23) and other deliverability expressions, the product is at the average pressure between Pe and Pwf. Temperature is constant. With the data in Appendix C, show the difference in the...
-
51. Prepare a datafile for the ADJUST program to solve the following horizontal network. T 2165.823 6963.374, 0.000 4148 349 4323.793 4558.156 Az 90d 3086.928 R 20 2587.737
-
1- Connect the circuit shown in Fig. 3. Use the NPN transistor BC107 V. +12V Re 470k www VB R BC107 Ve Figure 3: Practical Fixed Bias Transistor Circuit Transistor 1.(BC107) Quantity VB Vc VBEQ IBQ...
-
Why do most process-costing systems need to rely on an inventory cost flow assumption?
-
Selected condensed data taken from a recent statement of financial position of Morino Ltd. are as follows. MORINO LTD. Statement of Financial Position (partial) Other current assets...
-
Determine the half-cell reactions and the overall cell reaction, calculate the cell potential, and determine the equilibrium constant at 298.15 K for the cell Is the cell reaction spontaneous as...
-
Consider the half-cell reaction AgCl(s) + e Ag(s) + Cl (aq). If (AgCl, s) = 109.71 kJ mol 1 , and if E = +0.222 V for this half-cell, calculate the standard Gibbs energy of formation of Cl (aq).
-
For a given overall cell reaction, S o R = 16.5 J mol -1 K -1 and H o R = 270.0 kJ mol -1 . Calculate E o and (E o /t) P . Assume that n = 2.
-
Arlington Company has the following securities in its portfolio of trading equity securities on 12/31/23: 4,000 shares of Able Corp. 7,000 shares of Baxter Co. 2,500 shares of Connor Co. Cost...
-
The Shirt Shop had the following transactions for T-shirts for Year 1, its first year of operations: Jan. 20 Purchased 400 units $ 4 = $1,600 Apr. 21 July 25 Purchased 90 units @ $ 5 = 450 Sept. 19...
-
According to Paul Krugman, by each country, specializing in products for which it posses comparative advantage, countries could produce more in total and exchange products for goods that were cheaper...

Study smarter with the SolutionInn App


