Question: Some organic compounds containing the C=O group can react with themselves in a process known as aldol condensation. The mechanism for this reaction in acidic
Some organic compounds containing the C=O group can react with themselves in a process known as aldol condensation.
The mechanism for this reaction in acidic solution is shown here.
Write the overall reaction, identify any intermediates, and determine the role of the hydrogen ion.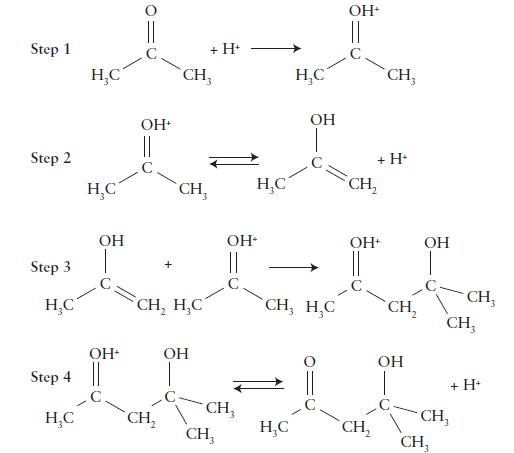
Step 1 Step 2 Step 3 Step 4 HC - www. OH || CH, ans CH, HC CH CH, + H+ OH - || CH, CH HC CH, H, C OH+ CH CH CH, + H CH2 OH OH CH CH CH, + H+ CH
Step by Step Solution
3.33 Rating (171 Votes )
There are 3 Steps involved in it
The image youve provided illustrates the steps of an aldol condensation mechanism in acidic conditions In this mechanism an aldehyde or ketone is acti... View full answer

Get step-by-step solutions from verified subject matter experts


