Suppose that 15.0 mL of 0.15 m NH 3 (aq) is titrated with 0.10 m HCl(aq). (a)
Question:
Suppose that 15.0 mL of 0.15 m NH3(aq) is titrated with 0.10 m HCl(aq).
(a) What is the initial pH of the 0.15 m NH3(aq)?
(b) What is the pH after the addition of 15.0 mL of 0.10 m HCl(aq)?
(c) What volume of 0.10 m HCl(aq) is required to reach halfway to the stoichiometric point?
(d) Calculate the pH at the halfway point.
(e) What volume of 0.10 m HCl(aq) is required to reach the stoichiometric point?
(f) Calculate the pH at the stoichiometric point.
(g) Use Table 6H.2 to select an indicator for the titration.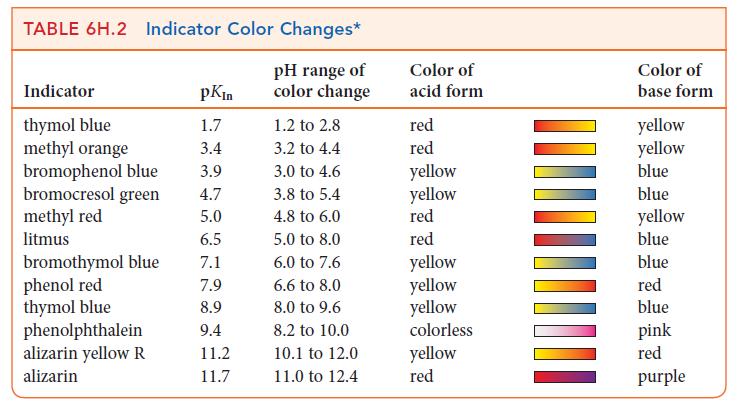
Transcribed Image Text:
TABLE 6H.2 Indicator Color Changes* pH range of color change Indicator thymol blue methyl orange bromophenol blue bromocresol green methyl red litmus bromothymol blue phenol red thymol blue phenolphthalein alizarin yellow R alizarin pKin 1.7 3.4 3.9 4.7 5.0 6.5 7.1 7.9 8.9 9.4 11.2 11.7 1.2 to 2.8 3.2 to 4.4 3.0 to 4.6 3.8 to 5.4 4.8 to 6.0 5.0 to 8.0 6.0 to 7.6 6.6 to 8.0 8.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 Color of acid form red red yellow yellow red red yellow yellow yellow colorless yellow red Color of base form yellow yellow blue blue yellow blue blue red blue pink red purple
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Initial pH 1120 b pH ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Suppose that 50.0 mL of 0.25 m CH 3 NH 2 (aq) is titrated with 0.35 m HCl(aq). (a) What is the initial pH of the 0.25 m CH 3 NH 2 (aq)? (b) What is the pH after the addition of 15.0 mL of 0.35 m...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
Below is the titration curve for the neutralization of 25 mL of a monoprotic acid with a strong base. Answer the following questions about the reaction and explain your reasoning in each case. (a) Is...
-
Northland Corporation is a small information-systems consulting firm that specializes in helping companies implement standard sales-management software. The market for Northalndss services is very...
-
The driver of a car traveling at 100 km/h toward a vertical cliff briefly sounds the horn. Exactly one second later she hears the echo and notes that its frequency is 840 Hz. How far from the cliff...
-
Ever since you walked through the door, your interviewee, Max Hugo, has been shuffling papers, looking at his watch, and drumming on his desk with his fingers. Based on what you know about...
-
Regional Support for Same Sex Marriage The website http://www.pewresearch.org/ fact-tank/2014/10/15/gay - marriage - arrives - in -the - south-where-the-public-is-less-enthused shows the changing...
-
Cash Flow to Creditors The 2006 balance sheet of Marias Tennis Shop, Inc., showed long-term debt of $3.1 million, and the 2007 balance sheet showed long-term debt of $3.3 million. The 2007 income...
-
Bristol Retail Group begins the year with inventory of $47,000 and ends the year with inventory of $37,000. During the year, the company has four purchases for the following amounts. Purchase on...
-
1. The Fisher Separation Theorem states that "If capital markets are complete and perfect, production decisions are governed solely by an objective market criterion, without regard to individuals'...
-
A metal alloy sample is believed to contain silver, bismuth, and nickel. Explain how it could be determined qualitatively that all three of these metals are present.
-
Calculate the pH of (a) 0.15 m CH 3 NH 3 Cl(aq); (b) 0.063 m FeCl 3 (aq).
-
A baseball analytics specialist wants to determine which variables are important in predicting a team's wins in a given season. He has collected data related to wins, earned run average (ERA), and...
-
Explain the difference between the overall government budget deficit, the current deficit, and the primary current deficit. Why are three deficit concepts needed?
-
You toss a \(0.40-\mathrm{kg}\) ball at \(9.0 \mathrm{~m} / \mathrm{s}\) to a \(14-\mathrm{kg}\) dog standing on an iced-over pond. The dog catches the ball and begins to slide on the ice. (a)...
-
Why do many governments have policies against negotiating with hostage-taking terrorists? Under what conditions, if any, are such policies likely to reduce hostage taking? Discuss the analogy to...
-
After a totally inelastic collision, the kinetic energy of an isolated system composed of two objects is zero. What was the momentum of the system in the same reference frame before the collision?
-
When two identical objects traveling at the same speed collide head-on, an observer standing in the Earth reference frame sees both objects changing direction. Do observers in every inertial...
-
A national survey showed that Hillshire Farm Deli Select cold cuts were priced, on the average, at $5.20 per pound. Suppose a national survey of 23 retail outlets was taken and the price per pound of...
-
(a) Bright Sdn Bhd (BSB) is a tax resident manufacturing company in Johor, which involves in ceramic tiles. Currently, BSBs annual sales turnover has been forecasted to be around RM 300,000 for the...
-
Can a gas be liquefied through an isenthalpic expansion if J T = 0?
-
Arrange the following compounds in order of increasing boiling point: -, - -, -0
-
Why is q V = U only for a constant volume process? Is this formula valid if work other than P V work is possible?
-
Kitt Ranch & Farm sells three grain/hay dryers to a local farmer at a total contract price of $48,000. The stand-alone price for each grain/hay dryer is $15,000 and the cost of each grain/hair dryer...
-
How do emergent paradigms in collaborative methodologies address the inherent complexities of modern research and development endeavors, particularly in the context of technological innovation and...
-
LemonWheel, Inc began 2023 with $6,296,053 in cash. Over the course of 2023, they experienced the following changes in their balance sheet. What is the ending cash balance for LemonWheel in 2023?...

Study smarter with the SolutionInn App


