Question: The wavefunction for a particle in a one- dimensional box is given in Eq. 2. Does the probability of finding the particle in the left-hand
The wavefunction for a particle in a one- dimensional box is given in Eq. 2. Does the probability of finding the particle in the left-hand one-third of the box depend on n ? If so, find the probability.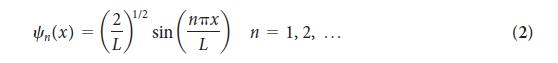
1/2 n(x) = (*) - () * sin(x) n = 1, 2, ... (2)
Step by Step Solution
3.51 Rating (164 Votes )
There are 3 Steps involved in it
The wavefunction youve provided describes a particle in a onedimensional box or an infinite potentia... View full answer

Get step-by-step solutions from verified subject matter experts


