Question: This plot shows the radial distribution function of the 3s- and 3p-orbitals of a hydrogen atom. Identify each curve and explain how you made your
This plot shows the radial distribution function of the 3s- and 3p-orbitals of a hydrogen atom. Identify each curve and explain how you made your decision.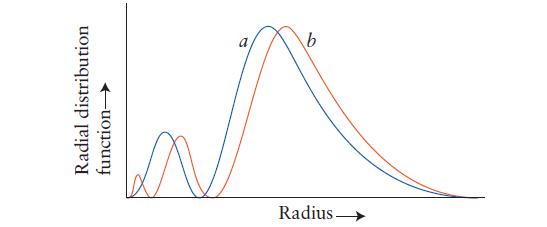
Radius Radial distribution function- b
Step by Step Solution
3.37 Rating (153 Votes )
There are 3 Steps involved in it
Use of a nonpolar solven... View full answer

Get step-by-step solutions from verified subject matter experts


