Use the information in Table 5G.2 to determine the value of K at 500 K for the
Question:
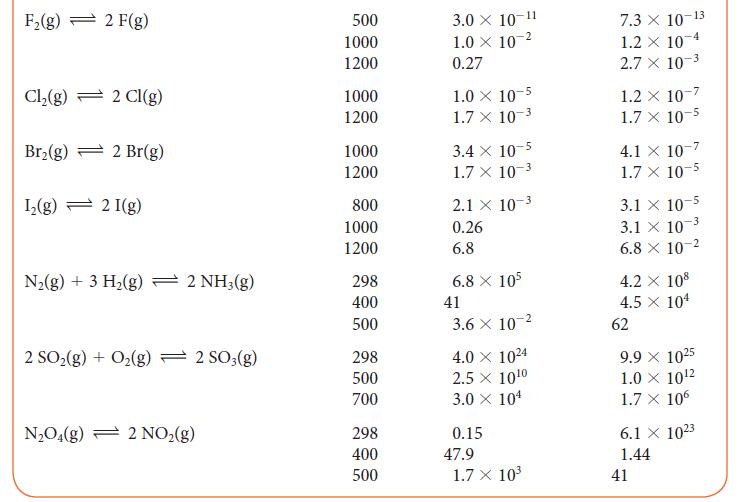
Use the information in Table 5G.2 to determine the value of K at 500 K for the reaction 2 NH3(g) + 3 I2(g) ⇌ N2(g) + 6 HI(g).
Transcribed Image Text:
TABLE 5G.2 Equilibrium Constants for Various Reactions Reaction H₂(g) + Cl₂(g) 2 HCl(g) H₂(g) + Br₂(g) → 2 HBr(g) H₂(g) + I₂(g) 2 HI(g) 2 BrCl(g) Br₂(g) + Cl₂(g) 2 HD(g) - H₂(g) + D₂(g) T/K* 300 500 1000 300 500 1000 298 500 700 300 500 1000 100 500 1000 K 4.0 × 10³1 4.0 × 10¹8 5.1 X 108 1.9 X 10¹7 1.3 × 10 ¹0 3.8 x 10¹ 794 160 54 377 32 5 0.52 0.28 0.26 K 4.0 × 10³1 4.0 × 10¹8 5.1 x 108 1.9 X 10¹7 1.3 × 10¹0 3.8 x 104 794 160 54 377 32 5 0.52 0.28 0.26
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
312g N2 6HI 2 NH31 Kc RTLog Ke AGAG AG products AG ran AG g products ...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
(a) In an experiment, 5.0 mmol Cl 2 (g) was sealed into a reaction vessel of volume 2.0 L and heated to 1200. K, and the dissociation equilibrium was established. What is the equilibrium composition...
-
Use the information in Table 5G.2 to determine the value of K at 300 K for the reaction 2 BrCl (g) + H 2 (g) Br 2 (g) + 2 HCl(g). TABLE 5G.2 Equilibrium Constants for Various Reactions Reaction H(g)...
-
Use the information in Table 2.5 to predict the standard reaction enthalpy of2 H2 (g) + 02(g) 2 H2O (1) at 100C from its value at 25C.
-
Apply Theorem 3 to calculate the matrix exponential e At for each of the matrices in Problems 35 through 40. A = 13 3 3 3 01 3 3 00 23 0002
-
Five hundred kmol/h of a liquid mixture of light alcohols containing, by moles, 40% methanol (M), 35% ethanol (E), 15% isopropanol (IP), and 10% normal propanol (NP) is distilled in a sequence of two...
-
Eventuallyand this may come sooner than most thinkthere will no longer be any choice locations anywhere in the world for new hamburger outlets. As a McDonalds stockholder, Im getting worried. Discuss.
-
Consider radiation exchange between two annular cylinders of radii \(R_{\mathrm{i}}\), and \(R_{\mathrm{o}}\) facing each other. This is similar to the radiation between flat plates considered in the...
-
(a) The distance to the North Star, Polaris, is approximately 6.44 x 1018 m. If Polaris were to burn out today, in what year would we see it disappear? (b) How long does it take for sunlight to reach...
-
A standardization battle is underway in the market for electric cars, with BMW, Nissan, Tesla and Toyota investing to establish their technology as the dominant standard. In the battle, Tesla has...
-
The Golden Oranges Nursery, which provides facilities for pre-school children on a commercial basis, is preparing its cash budget for next year. A profile of the estimated revenues and expenses for...
-
Two unknown molecular compounds were being studied. A solution containing 5.00 g of compound A in 100. g of water froze at a lower temperature than a solution containing 5.00 g of compound B in 100....
-
One step in the manufacture of sulfuric acid is the formation of sulfur trioxide by the combustion of SO 2 with O 2 in the presence of a vanadium(V) oxide catalyst. Suppose you are working out how to...
-
Discuss the role of the Systems Analyst in the preparation of the Business Case.
-
Jiangs best response to Subscriber 4 would be that active trading in trading in emerging market currencies: A. typically leads to return distributions that are positively skewed. B. should not lead...
-
Analyze the movement of the USD against the foreign currency for Portfolio A. Justify your choice. Kamala Gupta, a currency management consultant, is hired to evaluate the performance of two...
-
Given the recent movement in the forward premium for the SEK/EUR rate, Bjrk can expect that the hedge will experience higher: A. basis risk. B. roll yield. C. premia income. Rika Bjrk runs the...
-
Analyze the foreign-currency return for Portfolio B. Justify your choice. The fund manager of Portfolio B is evaluating an internally-managed 100% foreigncurrency hedged strategy. Kamala Gupta, a...
-
Describe two considerations for each type of component recommended to Grimmett for her manager selection process. Grimmett asks the adviser if any other preparatory steps should be taken before...
-
Set up a spreadsheet to solve Problem 6. In Problem 6, the construction company in Problem 2 decides to act as the owner on the project. The construction company plans to sell the project for...
-
The sales department of P. Gillen Manufacturing Company has forecast sales in March to be 20,000 units. Additional information follows: Finished goods inventory, March 1 . . . . . . . . . . . . . . ....
-
Propose a mechanism for the following transformation: 1) Excess LAH 2) H20 CH3
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
One liter of fully oxygenated blood can carry 0.18 liters of O 2 measured at T = 298 K and P = 1.00 atm. Calculate the number of moles of O 2 carried per liter of blood. Hemoglobin, the oxygen...
-
Assume you hold a 20-month to maturity bond, with 1,000 face value, semi- annual coupons of 7% p.a., interest rate to discount is 8% p.a. compounded semi- annually. What is the clean price of the...
-
Write equation in translation form. 10 f(4) = 6 0 -8 -6 61 18- 10 8 6 2 6 8 10
-
Interest rate risk and bond price changes Apex Corp. has two outstanding bond issues. One issue consists of 8% annual coupon bonds and the other issue consists of zero-coupon bonds. For each bond...

Study smarter with the SolutionInn App


