Propose a mechanism for the following transformation: 1) Excess LAH 2) H20 CH3
Question:
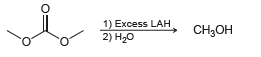
Transcribed Image Text:
1) Excess LAH 2) H20 CH3он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
JA HAH H ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a mechanism for the following transformation: NaOH, heat
-
Draw a mechanism for the following transformation: 'CI Z Z
-
Draw a mechanism for the following transformation: . HCI -
-
(2) Use Figure 10.2 shows a multi-degree-of-freedom (MDOF) model of three connected masses. Find the following for the MDOF system in Figure 10.2: Find expressions for the kinetic energy (T) and...
-
Quicksilver Delivery Service contracts to deliver Pete's Pizza Parlor's products to its customers for $5,000, payable in advance. Pete's pays the money, but Quicksilver fails to perform. Can Pete's...
-
Explain why organizations operating in certain environments often employ mechanistic structures.
-
Suppose that \(x_{i}\) only takes on the values 0 and 1. Out of the \(n\) observations, \(n_{1}\) take on the value \(x=0\). The \(n_{1}\) observations have an average \(y\) value of \(\bar{y}_{1}\)....
-
Jennifer Williamson recently received her MBA and has decided to enter the mortgage brokerage business. Rather than work for someone else, she has decided to open her own shop. Her cousin Jerry has...
-
The general ledger of Karlin Company, a consulting firm, as of January 1, 2018, contained the following account balances: Account Title Debits Credits Cash 30,200 Accounts Receivable 15,500 Equipment...
-
A production manager wants to assess the reactions of the blue-collar workers in his department (including foremen) to the introduction of computer-integrated manufacturing (CIM) systems. He is...
-
Show the reagents you would use to achieve the following transformation:
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
An S-590 alloy component (Figure 8.32) must have a creep rupture lifetime of at least 100 days at 500C (773 K). Compute the maximum allowable stress level.
-
HIM professions are employed in all areas that contribute to the revenue cycle. Each type of reimbursement has unique characteristics and a different approach to risk. List four reimbursement...
-
Is it possible to claim a deduction for childcare costs when both you and your spouse are employed full-time, with respective incomes of $80,000 and $40,000, and you employ a nanny for your...
-
Explaining how the provided statistical ratios are used in the financial services industry. ii. Explaining how the provided statistical analysis methods are used in the financial services industry....
-
With sales of $450,000, MJM, Inc. is operating at capacity but management anticipates that sales will grow 20 percent during the coming year. The company earns 8 percent on sales and distributes 50...
-
Find the regular function (analytic function) whose imaginary part 2-5 2442
-
When you view the Sun near the horizon, does refraction make it appear higher or lower in the sky?
-
Complete the equations for the following equilibria and calculate Keq where the Keq expression includes [HO]. Be sure to enter Keq in proper scientific notation. (a) ammonia (acting as a base) reacts...
-
Isoborneol (Problem 27.37) is converted into camphene on treatment with dilute sulfuric acid. Propose a mechanism for the reaction, which involves a carbocationrearrangement. C . H H2SO4 HO . H3...
-
Digit oxigenin is a heart stimulant obtained from the purple foxglove Digitalis purpurea and used in the treatment of heart disease. Draw the three-dimensional conformation of digitoxigenin, and...
-
What product would you obtain by reduction of digitoxigenin (Problem 27.39) with LiAlH4? By oxidation with pyridinium chlorochromate
-
A taxpayer disposed a part of asset for 100,000 SR in cash. The taxpayer purchased that asset for 90,000 SR 5 years ago but the market value of the complete assets was as follows: Part disposed...
-
A company maintains debt/equity ratio of 0.6. The flotation cost of equity is 12% and flotation cost of debt it is 6%. The firm is considering a new project which will require $5 million in external...
-
Flight Caf prepares in-flight meals for airlines in its kitchen located next to a local airport. The company's planning budget for July appears below: Flight Caf Planning Budget For the Month Ended...

Study smarter with the SolutionInn App


