Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m
Question:
Which of the following indicators in Table 6H.2 could you use for a titration of 0.20 m NH3(aq) with 0.20 m HCl(aq):
(a) Bromocresol green;
(b) Methyl red;
(c) Phenol red;
(d) Thymol blue? Explain your selections.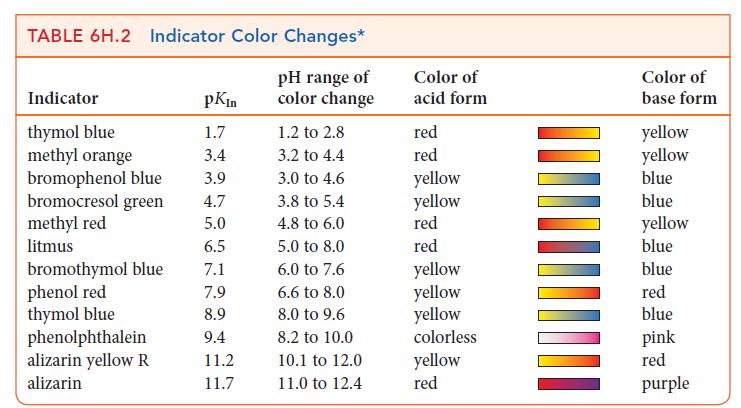
Transcribed Image Text:
TABLE 6H.2 Indicator Color Changes* pH range of color change Indicator thymol blue methyl orange bromophenol blue bromocresol green methyl red litmus bromothymol blue phenol red thymol blue phenolphthalein alizarin yellow R alizarin pKin 1.7 3.4 3.9 4.7 5.0 6.5 7.1 7.9 8.9 9.4 11.2 11.7 1.2 to 2.8 3.2 to 4.4 3.0 to 4.6 3.8 to 5.4 4.8 to 6.0 5.0 to 8.0 6.0 to 7.6 6.6 to 8.0 8.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 Color of acid form red red yellow yellow red red yellow yellow yellow colorless yellow red Color of base form yellow yellow blue blue yellow blue blue red blue pink red purple
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Given the pH at the equivalence point is approximately 237 thymol blue would be the most suitable ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
You would like to invest in one of the three available investment plans: money market, bonds, or stocks. The payoffs (profits) of each plan under two possible future economic conditions, PE (poor...
-
A series of titrations of lactic acid, CH 3 CH(OH)COOH (pK a = 3.86) is planned. About 1.00 mmol of the acid will be titrated with NaOH(aq) to a final volume of about 100 mL at the equivalence point....
-
Select indicators from Table 10-3 that would be useful for the titrations in Figures 10-1 and 10-2 and the pK a = 8 curve in Figure 10-3. Select a different indicator for each titration and state...
-
(a) A circular diaphragm 60 cm in diameter oscillates at a frequency of 25 kHz as an underwater source of sound used for submarine detection. Far from the source, the sound intensity is distributed...
-
Find the mass of a lead sphere of radius 2 cm.
-
What are the key problems involved in maintaining a competitive advantage in a growing industry environment?
-
For each of the following, calculate the degrees of freedom \((d f)\) and determine the critical values of \(t\) (assume \(\mathrm{a}=.05\) ). a. N =21, N2 = 21, H: H H1 H1* H2 b. N 14, N = 14, H1 H1...
-
Income statements of M Cop. and K Co. for the year ended December 31, Year 6, are presented below: Additional Information ¢ M Co. uses the equity method to account for its investment in K Co....
-
roblem Notification: 1. List the photos of things you would want to observe first hand upon being notified of the problem. Make this list in the Photos worksheet in the provided template. Problem...
-
Peyton Smith enjoys listening to all types of music and owns countless CDs. Over the years, Peyton has gained a local reputation for knowledge of music from classical to rap and the ability to put...
-
(a) What is the approximate chemical formula of rust? (b) What is the oxidizing agent in the formation of rust? (c) How does the presence of salt accelerate the rusting process?
-
(a) What must be the ratio of the molar concentrations of CO 3 2 and HCO 3 ions in a buffer solution having a pH of 11.0? (b) What mass of K 2 CO 3 must be added to 1.00 L of 0.100 m KHCO 3 (aq) to...
-
Explain why revenue recognition is a major problem in financial reporting.
-
A typical wind turbine extracts 40% of the kinetic energy of the wind that blows through the area swept by the blades. For a large turbine, 110,000 kg of air moves past the blades at 15 m/s every...
-
A 95 kg quarterback accelerates a 0.42 kg ball from rest to 24 m/s in 0.083 s. What is the specific power for this toss?
-
Male emperor penguins spend the long Antarctic night on the ice. They keep from freezing with exquisite thermal insulation. Their insulation is so effective that the surface temperature of their...
-
The spring in a retractable ballpoint pen is 1.8 cm long, with a 300 N/m spring constant. When the pen is retracted, the spring is compressed by 1.0 mm. When you click the button to extend the pen,...
-
A wind turbine works by slowing the air that passes its blades and converting much of the extracted kinetic energy to electric energy. A large wind turbine has 45-m-radius blades. In typical...
-
A survey was undertaken by Bruskin/Goldring Research for Quicken to determine how people plan to meet their financial goals in the next year. Respondents were allowed to select more than one way to...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
A balloon filled with 11.50 L of Ar at 18.7C and one atm rises to a height in the atmosphere where the pressure is 207 Torr and the temperature is 32.4C. What is the final volume of the balloon?...
-
Carbon monoxide competes with oxygen for binding sites on the transport protein hemoglobin. CO can be poisonous if inhaled in large quantities. A safe level of CO in air is 50. parts per million...
-
The total pressure of a mixture of oxygen and hydrogen is 1.65 atm. The mixture is ignited and the water is removed. The remaining gas is pure hydrogen and exerts a pressure of 0.190 atm when...
-
Using {blog} as your base, create words with the following meanings: A small blog Someone addicted to blogging A blog that begs A math-oriented blog A book based on a blog A blog devoted to...
-
Calculate the amount of capital gain on a $1000 corporate distribution to a shareholder if the shareholder share of earnings and profits is $600 and the shareholders stock basis is $200.
-
Find the values of x and y that satisfy the equation. 54-yi= 9x-4i and y

Study smarter with the SolutionInn App


